A clear, patient-friendly guide to whole genome sequencing, including how it works, what it reveals, and its role in diagnosis and research.
Understanding rare disease science and policy should not require a degree in genetics or medicine. The Explainer Library has been created to help every reader follow essential concepts with confidence. Each guide is short, plain language, and focused on one topic at a time. The aim is to remove confusion, reduce technical barriers, and support informed reading across all sections of Rare Disease Watch.
You will find clear explanations of topics such as the meaning of a rare disease, how genetic testing works, why some conditions run in families, how clinical research is designed, and what terms like prevalence, biomarkers, and endpoints actually signify in practice. These resources are written to support patients, families, researchers, students, and anyone curious about this complex field.
New explainers are added regularly and updated as understanding evolves. Readers can browse individual topics or search for the concepts they want to explore further.
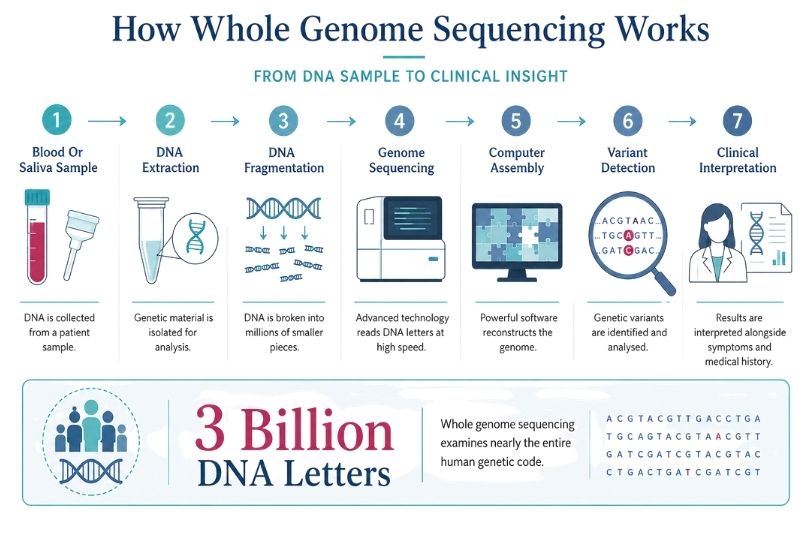 Whole Genome Sequencing: A Guide for Families
Whole Genome Sequencing: A Guide for Families
A clear, patient-friendly guide to whole genome sequencing, including how it works, what it reveals, and its role in diagnosis and research.
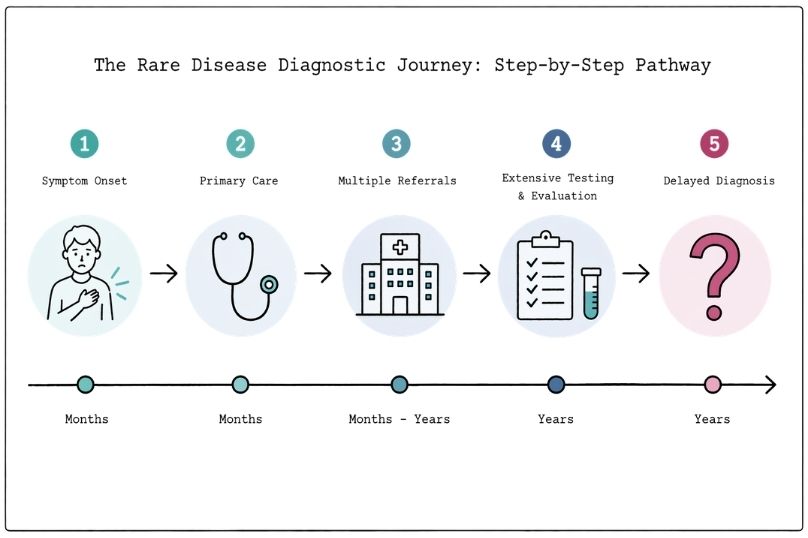 Understanding the Rare Disease Diagnostic Journey
Understanding the Rare Disease Diagnostic Journey
Rare disease diagnosis often takes years. Learn why delays occur, the barriers patients face, and how genomic testing is helping improve diagnosis.
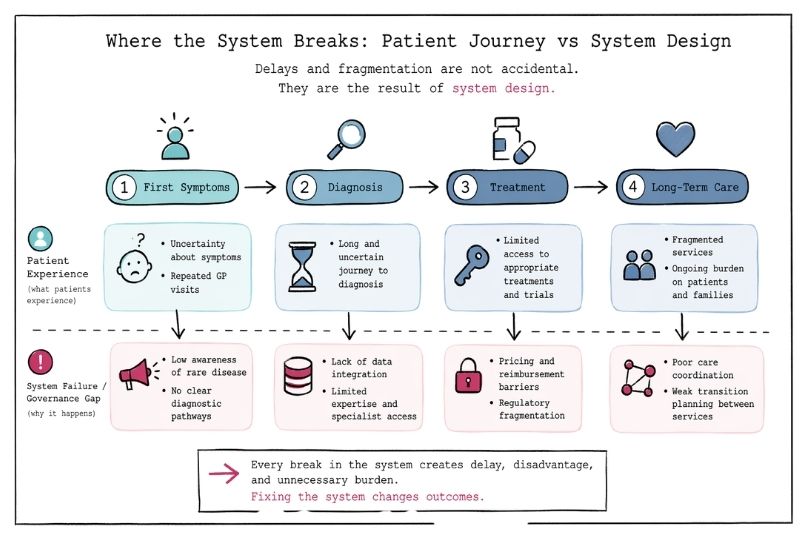 Rare Disease as a Systems Challenge: Policy, Data, and Care
Rare Disease as a Systems Challenge: Policy, Data, and Care
Explore rare disease as a systems challenge shaped by governance, data, and care design. See why long horizon, integrated approaches improve outcomes.
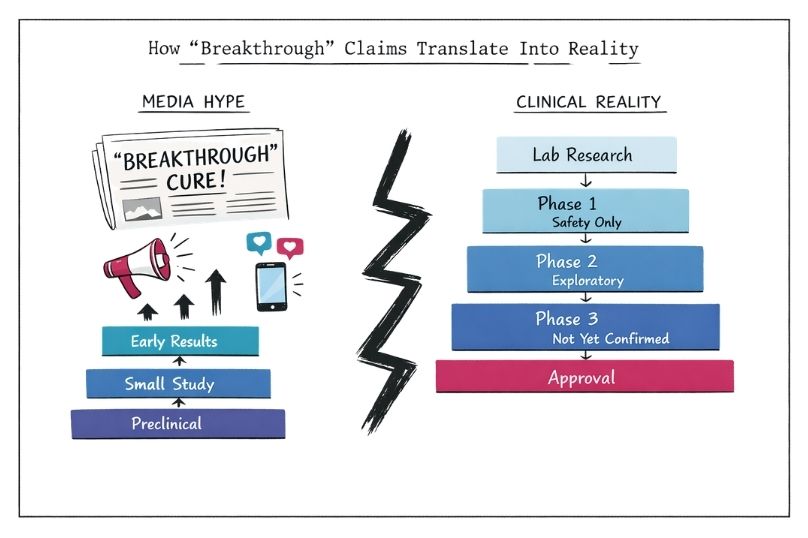 How to Interpret “Breakthrough” Treatment Claims in Rare Diseases
How to Interpret “Breakthrough” Treatment Claims in Rare Diseases
Learn how to critically assess “breakthrough” treatment claims in rare disease research. Understand evidence stages, avoid hype cycles, and set realistic expectations based on science.
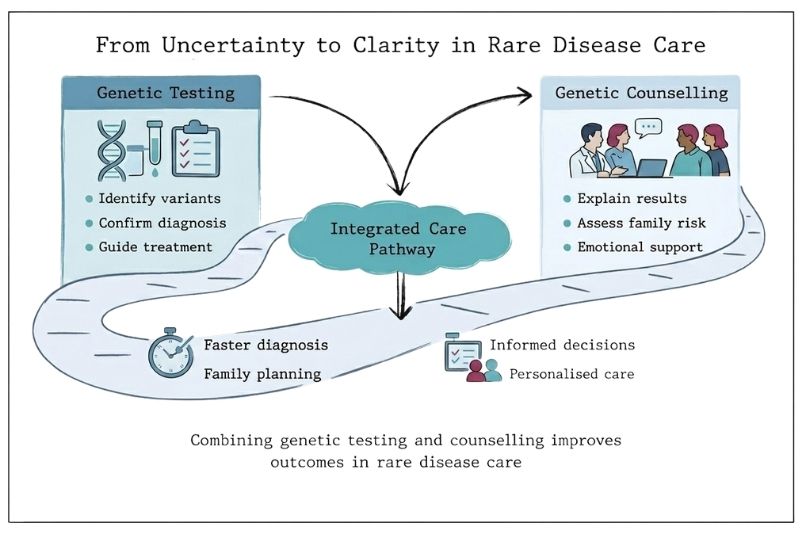 Reshaping Rare Disease Care: The Role of Genetic Testing and Counselling
Reshaping Rare Disease Care: The Role of Genetic Testing and Counselling
Learn how genetic testing and counselling work in rare diseases, and how this integrated approach supports earlier diagnosis and informed care.
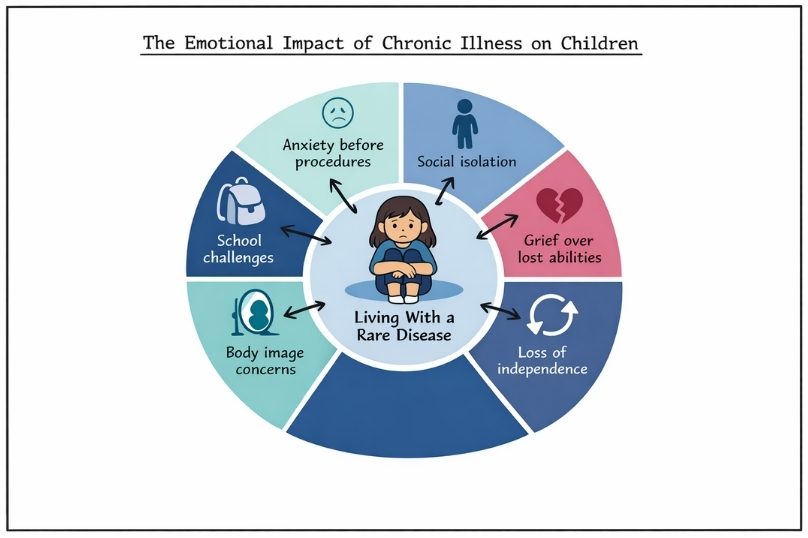 Supporting Your Child's Mental Health Through Chronic Illness
Supporting Your Child's Mental Health Through Chronic Illness
Support your child’s mental health through chronic illness with practical strategies to build resilience, encourage communication, and strengthen support systems.
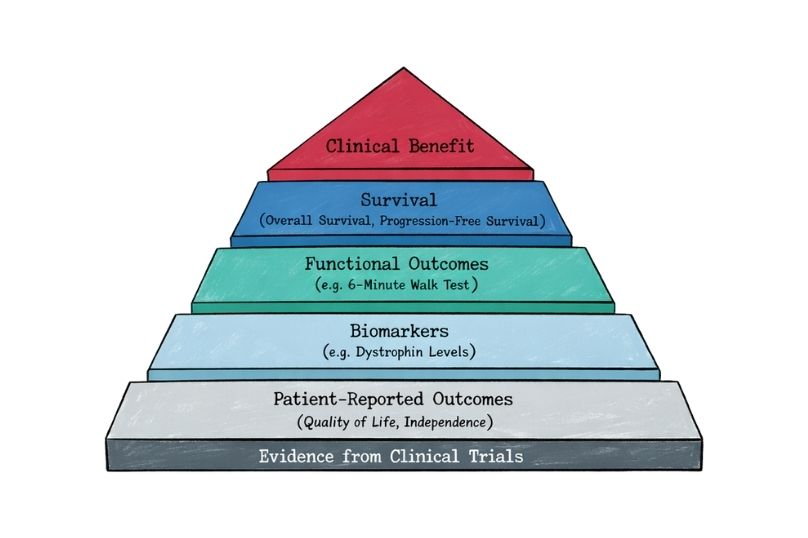 Endpoints in Rare Disease Clinical Trials
Endpoints in Rare Disease Clinical Trials
Explore how endpoints are defined in rare disease clinical trials, the challenges of small populations, and the growing role of patient-reported outcomes in regulatory decisions.
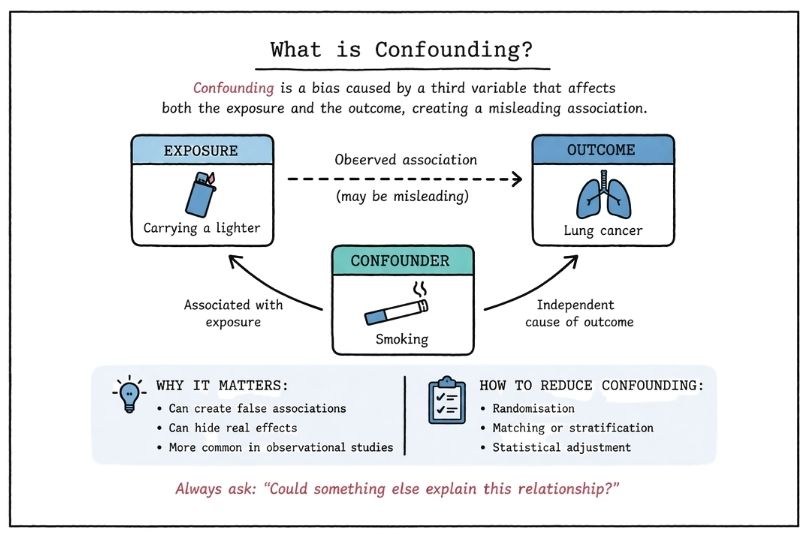 What Strong and Weak Evidence Mean in Medical Research
What Strong and Weak Evidence Mean in Medical Research
Understand strong vs weak evidence in medical research, including evidence hierarchy, bias, and expectation control. A clear guide with relevance to rare diseases and clinical decision-making.
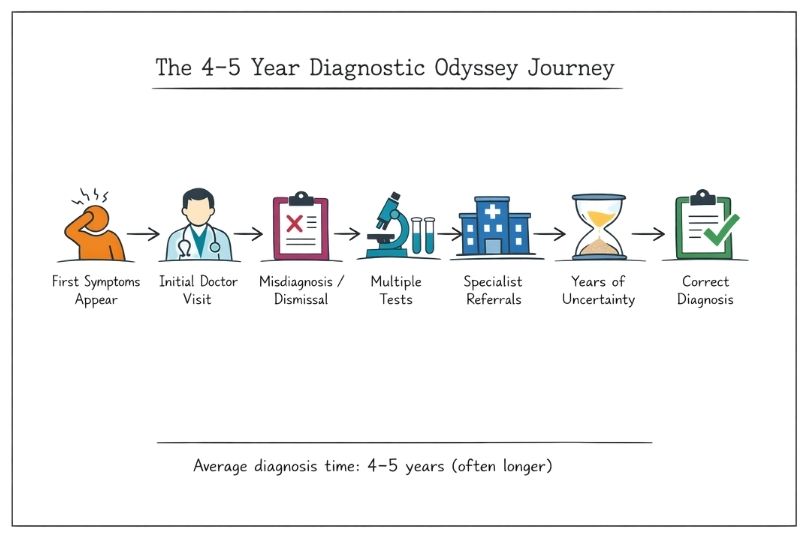 The Diagnostic Odyssey: Why Rare Diseases Are Hard to Identify
The Diagnostic Odyssey: Why Rare Diseases Are Hard to Identify
The diagnostic odyssey in rare diseases often takes 4–5 years. Discover why symptom overlap, misdiagnosis, system barriers, and genetic limits cause delays, plus emerging solutions.
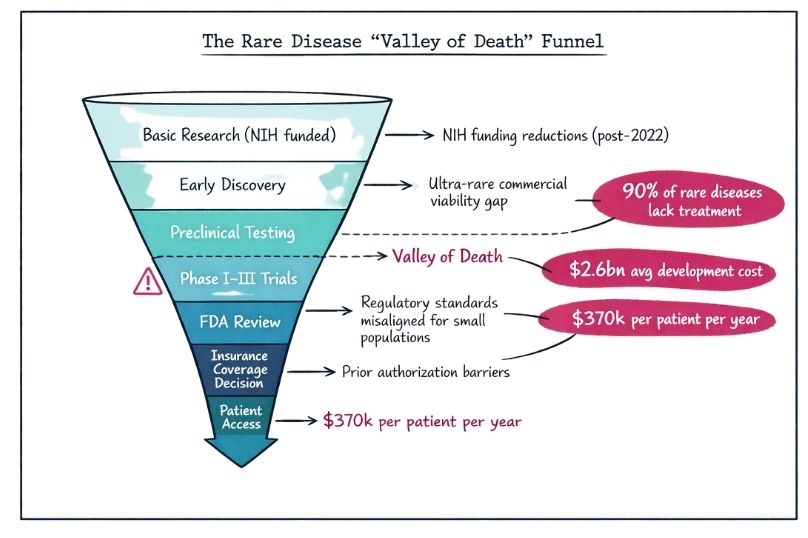 How Health Policy Shapes Access to Rare Disease Treatments
How Health Policy Shapes Access to Rare Disease Treatments
Explore shifts in rare disease policy and how they impact patient access to life-saving orphan drugs.