The language of medical progress is increasingly shaped by urgency. Headlines promise “game changing” discoveries, press releases describe “unprecedented” results, and early-stage findings are often framed as if they are ready to transform patient care. For individuals and families affected by rare diseases, this environment creates a difficult tension. On one hand, there is a genuine need for progress and a justified interest in emerging treatments. On the other, there is a growing risk that hope becomes entangled with hype.
Understanding how to interpret claims about “breakthrough” treatments is not about dampening optimism. It is about developing the literacy required to distinguish between early scientific signals and meaningful clinical impact. This distinction matters. It shapes expectations, informs decision-making, and protects individuals from cycles of excitement followed by disappointment.
This article provides a structured approach to evaluating such claims, with a particular focus on rare disease contexts where evidence is often limited, timelines are extended, and uncertainty is inherent.
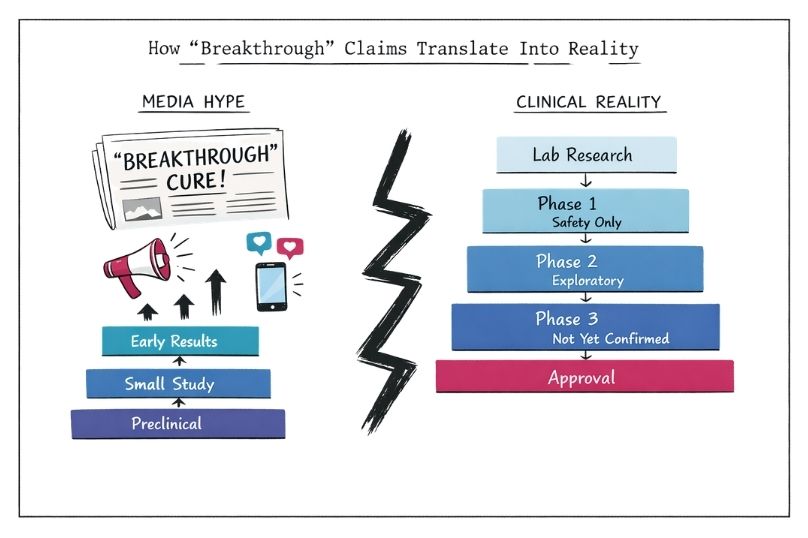
The term “breakthrough” has a specific meaning within regulatory and scientific frameworks, but it is frequently used more loosely in public communication.
In formal terms, a “breakthrough therapy designation” is granted by regulators such as the FDA or MHRA when early evidence suggests that a treatment may offer substantial improvement over existing options for serious conditions. This designation is not an approval. It is a mechanism to accelerate development and review.
In contrast, in media and marketing contexts, “breakthrough” is often used to describe:
These uses are not inherently misleading, but they can create a false sense of proximity to real-world treatment. The key issue is not the word itself, but the lack of clarity around what stage of development it actually refers to.
A useful starting point is therefore to treat the term “breakthrough” not as a conclusion, but as a prompt for further scrutiny.
To interpret any claim accurately, it is necessary to understand where a treatment sits within the broader development pathway.
Drug and therapy development is a staged process:
At each stage, the level of evidence increases, but so does the complexity and cost. Many treatments that show promise in early stages do not progress to later phases.
For rare diseases, this pathway is often more complex. Small patient populations make large trials difficult. Natural history data may be limited. Endpoints can be harder to define. As a result, early signals may carry more weight than they would in common conditions, which increases both their importance and the risk of overinterpretation.
When encountering a claim about a “breakthrough,” the first question should be: at which stage is this intervention?
A laboratory discovery is fundamentally different from a therapy that has completed Phase 3 trials. Treating them as equivalent leads to distorted expectations.
Scientific progress is incremental. Early studies are designed to detect signals, not to provide definitive proof.
A signal might include:
These are important. They justify further investigation. However, they do not establish that a treatment is effective in a clinically meaningful way.
Proof requires:
In rare diseases, the distinction between signal and proof is often compressed in public discourse. A small study showing improvement in a handful of patients may be presented as evidence of effectiveness. In reality, it is a starting point.
A disciplined approach is to ask: is this evidence exploratory, or confirmatory? Most “breakthrough” claims relate to exploratory findings.
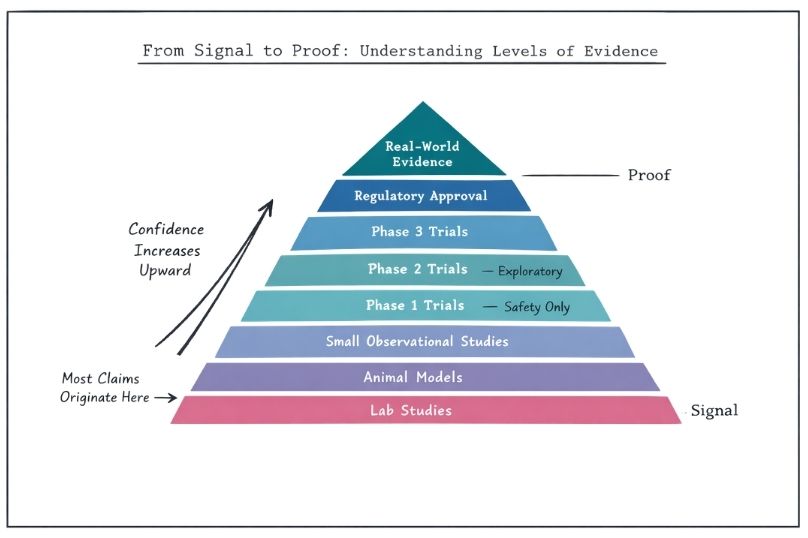
Not all studies carry the same weight. Understanding basic elements of study design can significantly improve interpretation.
Key considerations include:
Sample size
Small studies are more prone to variability and less reliable for generalisation
Control groups
Studies without a comparator make it difficult to attribute outcomes to the intervention
Randomisation
Without randomisation, selection bias can influence results
Blinding
If participants or researchers know who is receiving the treatment, expectations can affect outcomes
Endpoints
Surrogate endpoints, such as biomarker changes, may not translate into real clinical benefit
Duration
Short-term results may not reflect long-term safety or effectiveness
In rare disease research, compromises are often necessary due to practical constraints. However, these limitations should be recognised when interpreting claims.
A “breakthrough” based on an uncontrolled, short-term study in a small cohort should be viewed differently from one supported by a well-designed, multi-centre trial.
Many early claims are grounded in biological plausibility. A treatment targets a pathway believed to be central to the disease. In theory, this should produce benefit.
Biological plausibility is valuable. It guides research and helps prioritise interventions. However, it is not sufficient on its own.
Human biology is complex. Interventions that work in models do not always translate into clinical benefit. Pathways interact. Compensatory mechanisms exist. Side effects emerge.
For rare diseases, where understanding of underlying mechanisms may still be evolving, the gap between plausibility and outcome can be particularly wide.
When evaluating a claim, it is useful to distinguish between:
The former is a hypothesis. The latter is evidence.
Regulatory language can provide important context, but it is often misunderstood.
Terms such as:
Each has a specific meaning related to the development and review process.
For example, orphan designation recognises that a condition is rare and may provide incentives for development. It does not indicate effectiveness.
Conditional approval may allow a treatment to be used based on early evidence, with the requirement for further data. This reflects a balance between urgency and uncertainty.
Understanding these distinctions helps avoid overinterpreting regulatory milestones as indicators of clinical success.
The way research is communicated is shaped by incentives.
Academic institutions seek visibility and funding
Companies aim to attract investment and partnerships
Media outlets prioritise engagement and readership
This does not imply intentional misrepresentation. It reflects the reality that positive, forward-looking narratives are more likely to be amplified.
Press releases often highlight best-case interpretations. Limitations may be mentioned but not emphasised. Headlines simplify complex findings into accessible narratives.
For rare disease communities, this can create repeated cycles:
Over time, this pattern can erode trust or lead to fatigue.
A more sustainable approach is to engage with the underlying evidence rather than the framing.
One of the most important aspects of expectation management is understanding time.
Even under accelerated pathways, the development of new treatments typically spans years. For rare diseases, timelines can be longer due to recruitment challenges and the need for tailored study designs.
Early results do not translate into immediate availability. Manufacturing, regulatory review, reimbursement decisions, and clinical integration all take time.
When interpreting a “breakthrough,” it is therefore useful to consider:
This perspective does not diminish the importance of progress. It places it within a realistic framework.
For individuals living with rare diseases, scientific developments are not abstract. They are directly connected to quality of life, prognosis, and daily experience.
This context matters. It influences how information is received and interpreted.
Hope is not a problem. It is a rational response to limited options. However, when hope is repeatedly tied to early-stage findings that do not materialise into treatments, it can become destabilising.
A more constructive approach is to anchor hope in process rather than individual claims. Progress in understanding disease mechanisms, improving trial design, and building research infrastructure all contribute to long-term outcomes.
Interpreting “breakthrough” claims through this lens supports resilience without disengagement.
A structured set of questions can help assess any claim:
This framework does not require specialist training. It requires consistency.
Over time, applying these questions builds intuition and reduces susceptibility to hype cycles.
A common challenge in interpreting medical claims is the tendency towards binary conclusions. A treatment is seen as either a success or a failure.
In reality, most developments fall somewhere in between.
Recognising this nuance is particularly important in rare diseases, where heterogeneity is common and personalised approaches may be necessary.
Avoiding binary thinking allows for a more accurate and less emotionally volatile engagement with emerging evidence.
Interpreting complex medical information is demanding. Trusted intermediaries play a critical role.
These may include:
Their value lies in contextualising findings, highlighting limitations, and providing balanced interpretation.
However, even trusted sources should be evaluated critically. Transparency around evidence, uncertainty, and potential conflicts of interest remains essential.
At a broader level, the challenge is not simply to correct individual misunderstandings. It is to build a culture in which informed engagement with medical information is the norm.
This involves:
For rare diseases, where information gaps are common, this culture is particularly important.
It enables individuals to engage with developments without being driven by cycles of hype and disappointment.
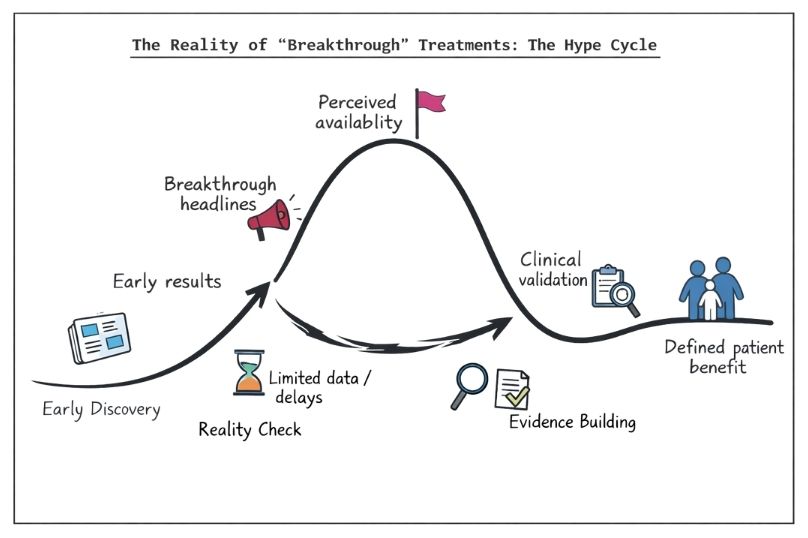
Claims about “breakthrough” treatments are a visible and often compelling part of the medical landscape. They reflect genuine effort, innovation, and progress. However, they also require careful interpretation.
Understanding where a treatment sits in the development pathway, distinguishing between signal and proof, evaluating study design, and recognising the influence of framing all contribute to a more accurate assessment.
For rare disease communities, this approach is not about reducing optimism. It is about aligning expectations with evidence. It supports better decision-making, more stable engagement with research developments, and a clearer view of what progress actually looks like.
Progress in medicine is real, but it is rarely instantaneous. Interpreting it with precision allows that progress to be recognised without distortion.