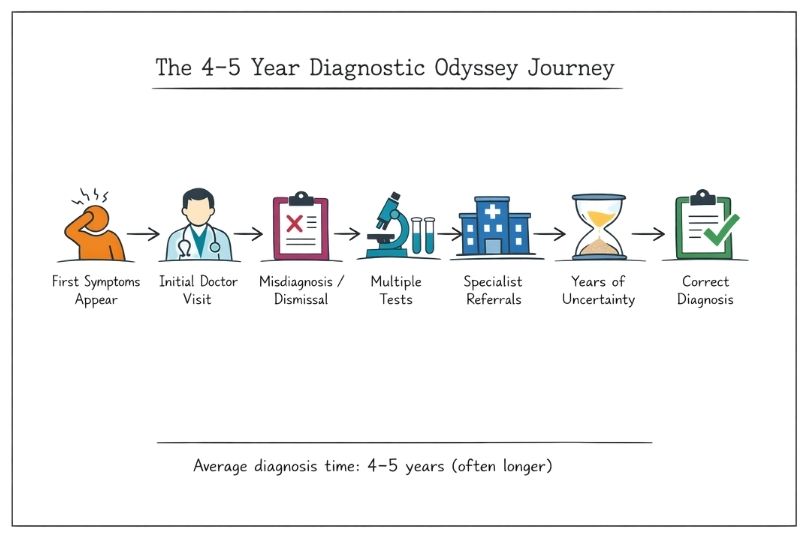
The “diagnostic odyssey” is one of the most challenging aspects of rare disease care. It refers to the prolonged and complex journey patients face, which often involves multiple consultations from the first appearance of symptoms to finally receiving an accurate diagnosis. Affecting millions of people worldwide, this process typically takes an average of 4–5 years, according to the Rare Barometer survey, although many patients wait significantly longer. The delay is more than frustrating; it brings years of uncertainty, repeated doctor visits, missteps, and a relentless search for answers before the condition is properly identified.
For the estimated 300 million people worldwide living with rare diseases, these long delays carry serious consequences. Patients endure prolonged suffering, receive inappropriate treatments, face mounting medical costs, and experience deep psychological distress. Each year without a clear diagnosis adds emotional strain, erodes trust in healthcare, and reduces quality of life. Understanding why rare diseases are so difficult to identify is the first step toward creating faster, more effective diagnostic pathways that can deliver timely answers and better outcomes for millions.
One of the fundamental obstacles in diagnosing rare diseases is that their symptoms often look exactly like more common conditions. Healthcare professionals are trained to consider the most likely explanations first, following the principle that “common things are common.” While logical, they naturally start with the usual diagnoses, which is reasonable, but this approach often delays recognition of rare diseases.
A EURORDISCare survey found that 40% of rare disease patients were initially misdiagnosed, leading to serious consequences:
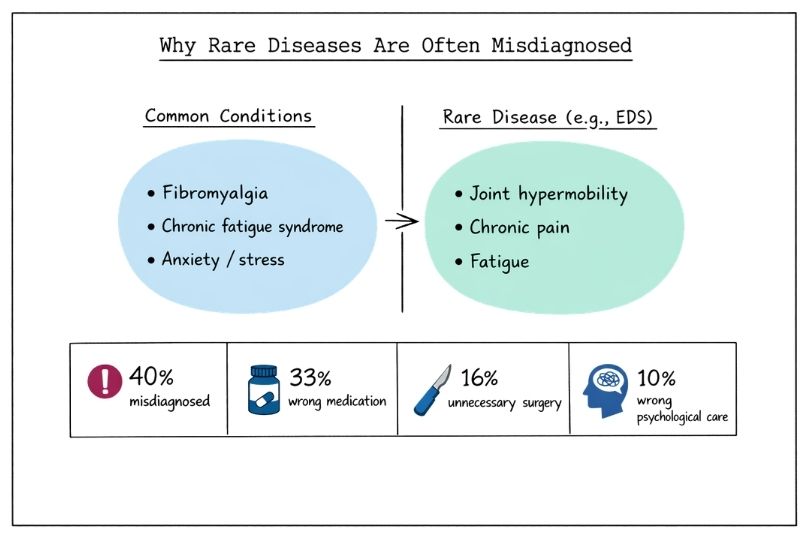
Ehlers-Danlos Syndrome (EDS) is a clear example. People with EDS often have joint hypermobility, chronic pain, and fatigue, symptoms that are frequently mistaken for fibromyalgia, chronic fatigue syndrome, or other common musculoskeletal conditions. As a result of mismanagement, many are told it's "just stress" or "growing pains" for years before the correct diagnosis is made.
The situation becomes even more complex when considering that many rare diseases present with non-specific symptoms such as fatigue, pain, developmental delays, or neurological issues. These signs are common across numerous conditions, making it extremely difficult for clinicians to suspect a rare disease without specialized knowledge or a high level of suspicion. As a result, patients often endure prolonged diagnostic journeys, highlighting the urgent need for greater awareness, improved training, and more refined diagnostic tools to shorten the path to accurate identification.
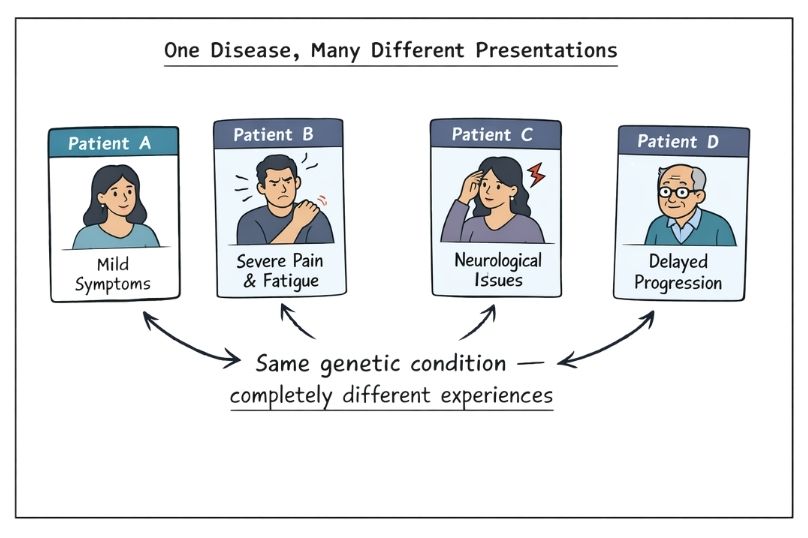
Rare diseases are often hard to diagnose because of their clinical heterogeneity; the same condition can look very different from one patient to another. Awareness does not always translate into understanding, even when doctors know a condition exists; its variability in symptoms, severity, and progression can make recognition extremely difficult. This variability means that the "classic" or textbook presentation taught in medical training is usually the exception rather than the rule, making accurate identification much harder in real-world practice.
Another major barrier is the limited exposure most healthcare professionals have to rare diseases. Even specialists may encounter only a handful of cases throughout their entire career. With more than 7,000 known rare diseases, it’s impossible to be familiar with them all. This lack of regular experience creates a significant knowledge gap where clinicians don’t have sufficient experience to recognize atypical presentations, subtle clues, or unusual patterns that don’t match what they’ve seen before.
The challenge goes beyond individual doctors. Many rare diseases still lack clear, established diagnostic protocols or standardized treatment guidelines. When a patient presents with unusual or inconsistent symptoms, healthcare providers may struggle to recognize what researchers call "pattern failures", situations where recurring symptoms or unexpected features should trigger a re-evaluation of the initial diagnosis. Without clear frameworks or repeated exposure, it’s easy for important signals to be overlooked.
This experience gap is especially noticeable in primary care settings, where most patients first seek medical help. Primary care physicians have broad training and see a wide range of common conditions, but cannot reasonably be expected to stay current on thousands of rare diseases and their many variations. This often means the first doctor a patient sees may not suspect a rare condition; as a result, many patients face prolonged diagnostic journeys before reaching the right specialist or receiving an accurate diagnosis.
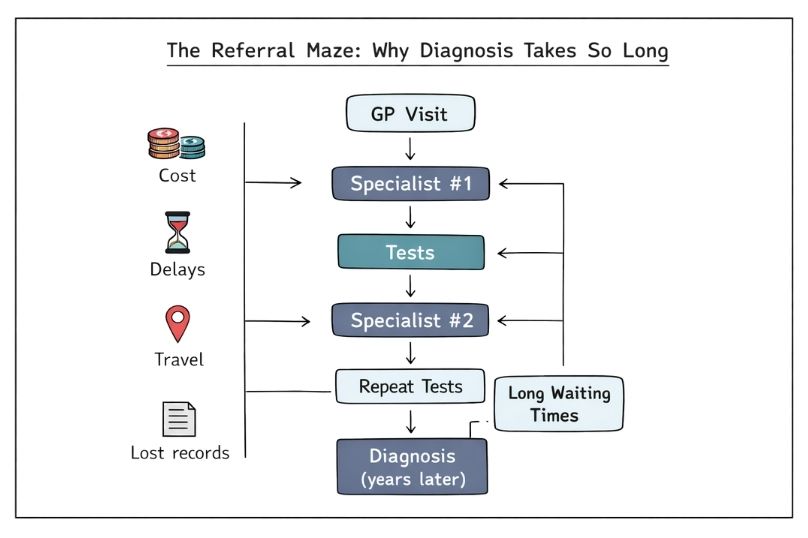
Modern healthcare systems are not designed with rare diseases in mind. Specialist expertise for rare conditions is mostly concentrated in large urban hospitals, academic medical centers, and major cities, leaving many geographic regions underserved and millions of patients with very limited or no access to the right kind of care, forcing people to travel long distances or accept long waits to see someone who might understand their condition.
This geographic inequality creates several practical and financial barriers:
Access Limitations:
At the same time, fragmented care pathways add further delays: patients are shuffled between multiple referrals, providers fail to communicate effectively, tests are repeated unnecessarily, and medical records are often incomplete or lost.
The referral process itself can be a major source of delay. A patient typically starts with a primary care physician, who refers them to one specialist, who may then refer them to another, and so on. Each referral can take weeks or months, especially when specialists have long waiting lists or limited availability. This drawn‑out process prolongs uncertainty and slows access to an accurate diagnosis.
Additionally, the healthcare system’s focus on evidence‑based medicine, while valuable in most cases, can be disadvantageous to rare disease patients. Because rare diseases affect small populations and have limited research, evidence‑based guidelines are often unavailable or insufficient. Clinicians may therefore lack the tools to make informed decisions, leaving patients without clear diagnostic or treatment pathways.
Despite major advances in genomic medicine, genetic testing still comes with significant challenges in diagnosing rare diseases. While genetic technologies have greatly expanded our understanding of rare diseases and made many diagnoses possible, their immediate clinical utility remains limited by factors such as uncertain variant interpretation, incomplete genetic knowledge, and the complexity of linking results to clear diagnostic outcomes.
Testing Access and Utilization:
Even with powerful genetic testing tools now available, they are still not used nearly enough. Currently, fewer than 10% of individuals undergo genetic screening, even among high-risk groups like critically ill children in intensive care units. Recent studies show that genetic testing remains significantly underutilized, despite strong clinical guidelines and expert recommendations advocating for increased genetic testing in appropriate cases.
Technology Limitations:
Many healthcare systems continue to rely on older or lower-resolution genetic testing technologies, such as:
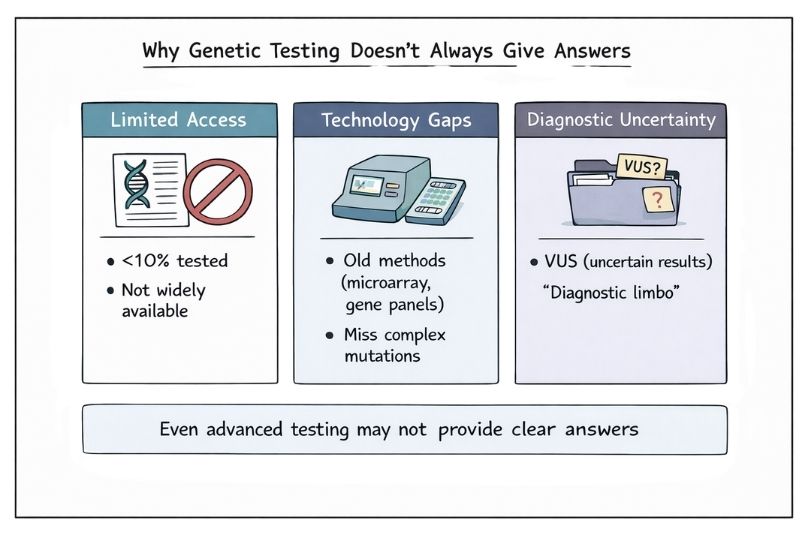
While these methods can detect certain genetic conditions, they generally have much lower diagnostic success rates compared to comprehensive genomic approaches like whole-exome or whole-genome sequencing. As a result, they often miss the underlying cause when the condition involves multiple genes, complex variants, or non-coding regions, leaving patients with incomplete or negative results even after testing. This reliance on limited tools contributes to prolonged uncertainty and repeated rounds of testing for many rare disease patients.
Variants of Uncertain Significance (VUS):
Even when comprehensive genetic testing is performed, the results can include variants of uncertain significance (VUS). These are genetic changes that cannot yet be clearly classified as harmful (pathogenic) or harmless (benign), leaving patients and clinicians in a frustrating state of “diagnostic limbo” with no definitive answer about whether the variant is causing the disease. In many cases, determining whether a variant is causally linked to disease requires further research and validation. See how researchers confirm a link between a gene and a disease in our guide to gene discovery.
While some VUS may eventually be reclassified as the genetic databases grow and more research becomes available, this process often takes years. In the meantime, it provides little immediate clarity or guidance for patients and families who are desperately seeking answers and appropriate care. This uncertainty adds another layer of emotional strain to an already difficult diagnostic journey.
Interpretation Challenges:
Genetic testing results are often complex and require specialized expertise to interpret correctly. Many healthcare providers, including general practitioners and even some specialists, lack the advanced training needed to analyze detailed genetic reports or to explain their implications clearly to patients. This gap in expertise can lead to confusion and miscommunication, which adds further delays to the diagnostic process, leaving patients without the clear guidance they need.
The prolonged diagnostic odyssey takes a heavy toll on patients’ psychological well-being and overall quality of life. The constant uncertainty, endless search for validation, and lack of clear answers create a significant emotional burden that affects not only the patients but also their families. This prolonged wait often leads to feelings of doubt, frustration, helplessness, and isolation, making daily life feel heavier and more difficult than it already is.
Patient Experience During Diagnosis:
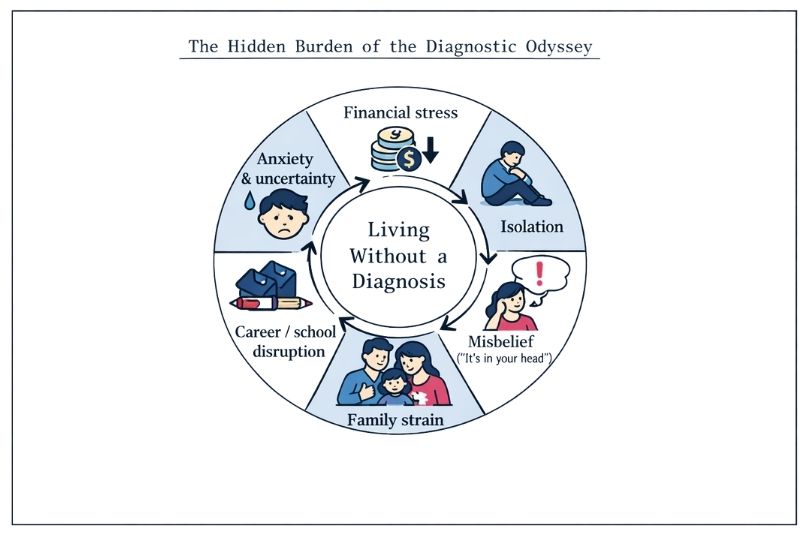
The psychological impact of the diagnostic odyssey reaches far beyond individual patients, often affecting entire family systems. Parents of children with undiagnosed conditions often carry heavy feelings of guilt, helplessness, and chronic stress, wondering if they missed something or could have done more. The ongoing uncertainty can strain relationships, disrupt family dynamics, and create tension, especially when symptoms are invisible or not understood by friends, extended family, or even some healthcare providers.
Many patients also describe feeling dismissed or disbelieved by healthcare providers, especially when early test results appear normal or inconclusive. Being told “it’s all in your head,” “you’re just anxious,” or “the tests are fine” is common and deeply hurtful. This lack of validation can erode trust, make people reluctant to keep seeking help, avoid follow-up appointments, or struggle to advocate for themselves, leaving them feeling even more isolated and unheard during an already difficult time.
Secondary Health Consequences:
The long delay in getting a correct diagnosis doesn’t just cause frustration; it can lead to serious secondary health problems that make the condition even harder to manage. These consequences often include:
The growing recognition of the challenges posed by the diagnostic odyssey has driven the development of several promising strategies aimed at improving the identification and diagnosis of rare diseases. These innovations, spanning technology, clinical practice, and collaborative care, offer hope for reducing delays and easing the burden on patients and families.
Genomic Medicine Advances:
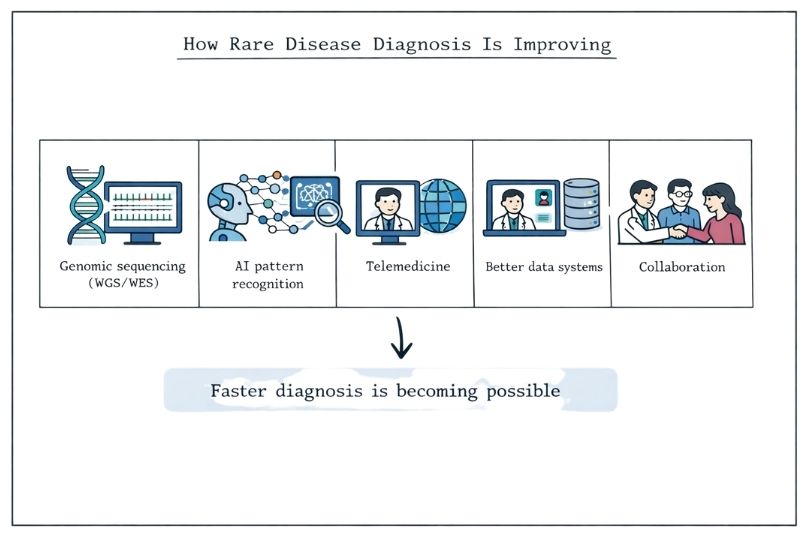
One of the most promising developments is the shift toward early and comprehensive genomic testing, particularly whole-genome sequencing (WGS) and whole-exome sequencing (WES). These advanced technologies allow doctors to examine a patient’s entire genetic code (or the most relevant parts of it) right at the start of the diagnostic process. They can quickly and accurately identify many genetic disorders that would otherwise take years to uncover through traditional step-by-step testing.
Although WGS and WES are still relatively expensive, the investment often pays off in the long run. FDA's new breakthrough pathways are also helping accelerate access to treatments for rare conditions. By providing a rapid diagnosis, these genomic tests can help avoid unnecessary repeat investigations, shorten hospital stays, prevent inappropriate treatments, and enable earlier access to the right therapies or management strategies. Not only does this reduce overall healthcare costs, but it also spares patients and families from prolonged uncertainty and suffering. As sequencing costs continue to fall and availability increases, these tools deliver both clinical and economic benefits while improving patient outcomes.
Technology Tools:
Collaborative Approaches:
One of the most promising ways forward is stronger collaboration across different fields. Bringing together geneticists, clinicians, researchers, and patient advocates in a true interdisciplinary way improves our overall understanding of rare diseases and leads to better management and care.
Patient advocacy organizations play an important role in this process. They connect families, help fund critical research, provide emotional and practical support, and raise awareness among healthcare providers. Resources such as the NORD directory of patient organizations and multidisciplinary clinics for rare diseases help bridge the gap between patients, families, and specialized professionals. These groups help drive faster recognition, more accurate diagnosis, and more compassionate treatment for everyone affected by rare conditions.
Educational Initiatives:
Medical education programs are placing greater emphasis on rare disease awareness and effective diagnostic approaches. Programs now include more training on recognizing unusual symptom patterns, knowing when to suspect a rare condition, and understanding the value of early specialist referral or genetic testing. For practicing physicians, NORD’s Continuing Medical Education (CME) courses on rare diseases and other initiatives help bridge existing knowledge gaps and improve recognition of rare disease presentations. These efforts help ensure that clinicians are better equipped to identify atypical symptoms and pursue appropriate diagnostic pathways.
The diagnostic odyssey remains a complex challenge that demands systemic change. As our knowledge of rare diseases grows and diagnostic technologies like genomic sequencing become more accessible and affordable, the possibility of significantly reducing these long delays is becoming realistic and achievable. Achieving this goal depends on sustained collaboration between patients and families, healthcare providers, researchers, and policymakers to build better systems, faster pathways, and more supportive environments for diagnosis.
For healthcare systems worldwide, addressing the diagnostic odyssey is both an urgent priority for patient care and an opportunity to create more efficient, patient-centered approaches to complex medical diagnosis. The way forward requires ongoing commitment to raising awareness for rare diseases, expanding access to specialized expertise, and leveraging technological advances to benefit the millions of patients currently navigating this challenging journey.