Rare disease is often framed as a clinical problem. In practice, it is a systems problem. The defining features of rare conditions, low prevalence, diagnostic uncertainty, fragmented expertise, and limited commercial incentives, expose structural weaknesses across health systems. These weaknesses are not confined to any single country or care pathway. They sit at the intersection of policy design, data infrastructure, regulatory frameworks, and service delivery.
Addressing rare disease effectively requires a shift in perspective. It is not sufficient to optimise individual components such as diagnostics or drug development in isolation. The challenge is to design systems that can operate coherently over long time horizons, under conditions of uncertainty, and at relatively small scale per condition but large scale in aggregate. This is fundamentally a governance question.
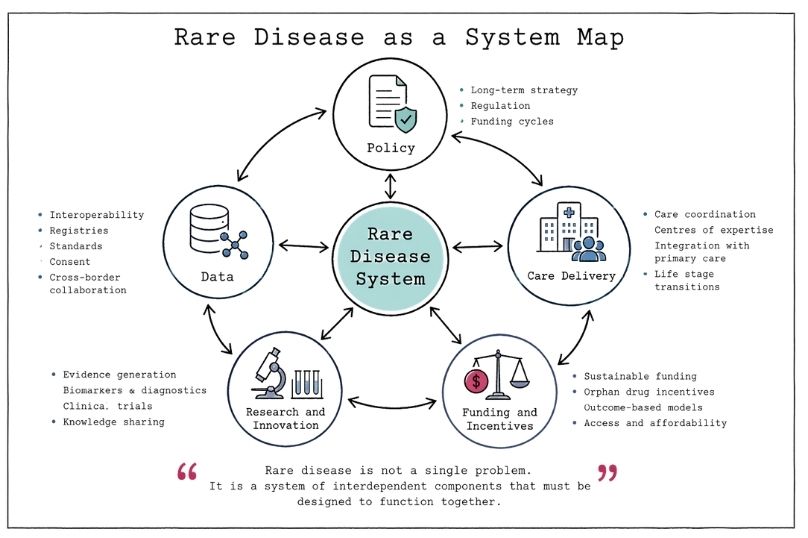
Rare diseases collectively affect hundreds of millions of people worldwide, despite each individual condition affecting a small number of patients. This creates a paradox. At the system level, rare disease is not rare. At the operational level, each condition lacks scale.
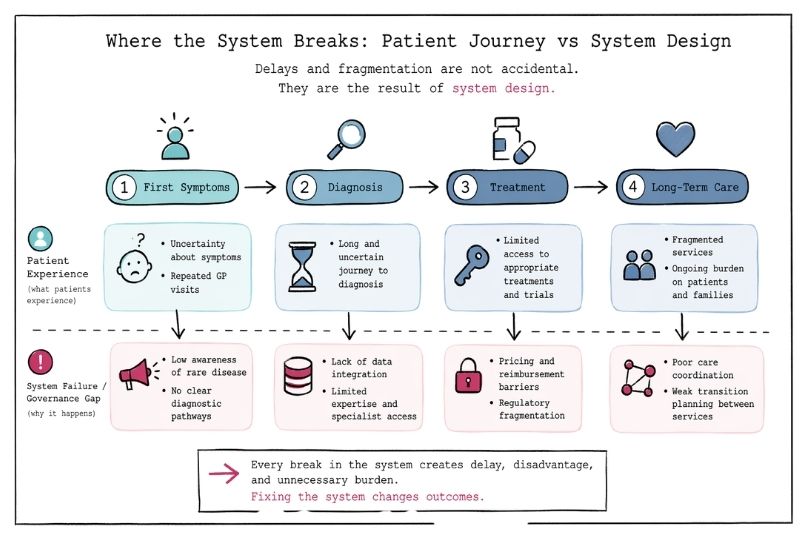
This mismatch produces predictable consequences. Delayed or missed diagnosis due to limited clinical familiarity. Fragmented care pathways across specialties and institutions. Inconsistent access to treatments and clinical trials. Weak data aggregation and interoperability. Limited commercial incentives for therapeutic development.
These are not isolated failures. They are system design outcomes.
Health systems have historically been optimised for high-prevalence conditions, where standardisation, economies of scale, and predictable demand support efficiency. Rare diseases do not conform to this model. They require flexibility, coordination, and sustained investment in areas where immediate returns are not always visible.
In this context, rare disease becomes a stress test of system maturity. It reveals whether governance structures can handle complexity, uncertainty, and long-term value creation.
Policy frameworks for rare diseases often focus on specific interventions such as orphan drug designation, funding mechanisms, or national rare disease strategies. These are necessary but insufficient.
A recurring limitation is temporal misalignment.
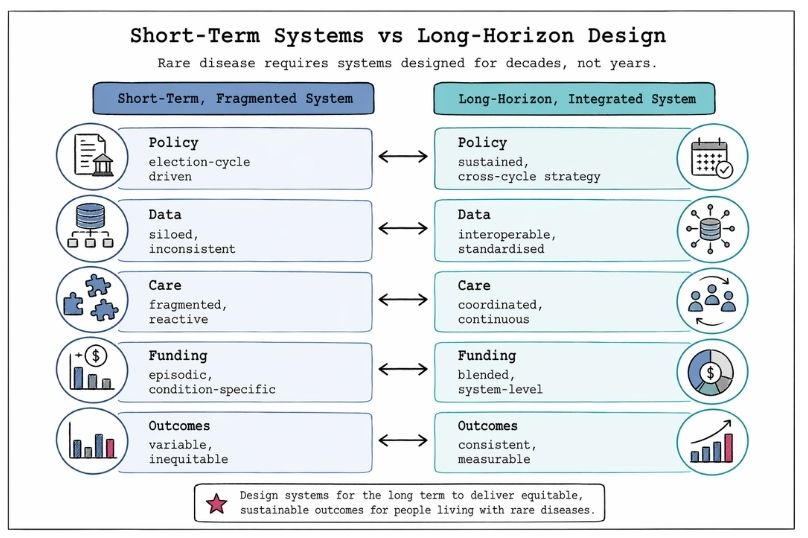
Policy cycles tend to operate on short- to medium-term horizons, driven by political and budgetary constraints. Rare disease requires long-horizon thinking. Diagnostic journeys can span years. Therapeutic development can take decades. Care needs are lifelong.
This creates a structural gap between policy timelines and lived reality.
Effective governance requires continuity across policy cycles so that strategies persist beyond electoral timelines, ideally with cross party support. It requires integrated policy design where health, social care, research, and data policy align rather than operate in silos. It requires outcome based frameworks that prioritise long term patient outcomes rather than short term activity metrics. It also requires adaptive regulation that can accommodate evolving scientific understanding, particularly in genomics and precision medicine.
Without this alignment, even well-intentioned policies risk becoming fragmented interventions that fail to scale or sustain impact.
Data is central to rare disease, not as an adjunct but as a core infrastructure.
For many conditions, clinical knowledge is sparse. Individual clinicians may see only a handful of cases in their careers. Progress depends on the ability to aggregate data across institutions, regions, and countries.
This introduces several governance challenges.
Interoperability remains a persistent barrier, with health data systems fragmented and lacking standardisation. Rare disease requires systems that allow data to be shared and analysed at scale while preserving clinical nuance.
Data quality and standardisation are equally critical. Inconsistent coding, variable diagnostic criteria, and incomplete records reduce the utility of datasets. Governance frameworks must define standards that enable aggregation without oversimplifying complexity.
Consent and trust must be actively managed. Patients are often willing to contribute data, but this depends on transparency, clear consent processes, and confidence that data will be used responsibly.
Cross-border collaboration is essential. Given the low prevalence of individual conditions, international data sharing is required to generate meaningful insights. This demands alignment of regulatory frameworks, ethical standards, and data sharing agreements.
Data governance in this context is not simply about compliance. It is about enabling cumulative knowledge that translates into improved outcomes.
Care for rare disease patients is frequently characterised by fragmentation. Patients often move between specialists, institutions, and services without a clear coordinating structure.
This reflects a systems design issue rather than a purely clinical one.
There is a need for defined coordination models, whether through dedicated care coordinators, multidisciplinary teams, or structured referral pathways. Centres of expertise can improve outcomes by concentrating knowledge, but they must be balanced with accessibility for patients who cannot easily travel.
Integration with primary care remains limited. Strengthening this interface can improve continuity and reduce the burden on patients to navigate complex systems independently.
Transitions across life stages are a known point of failure. Many rare diseases are lifelong, and movement from paediatric to adult services often results in discontinuity. Governance frameworks must explicitly address these transitions to ensure consistency of care.
These are structural design decisions that determine whether care is coherent or fragmented.
The development of therapies for rare diseases has been supported by policy mechanisms such as orphan drug designation, which provide incentives including market exclusivity and regulatory support.
These mechanisms have delivered progress but also expose limitations.
High development costs relative to small patient populations often lead to high pricing, creating access challenges. Commercial incentives may favour certain conditions or technologies over others. Post-approval evidence generation is frequently limited, particularly for ultra-rare conditions.
This raises questions about the balance between market mechanisms and public or philanthropic investment.
A more balanced approach may include blended funding models combining public, private, and philanthropic capital. Outcome-based reimbursement models may be appropriate in certain contexts. Investment in platform technologies that can address multiple conditions can improve efficiency. Support for non-commercial research pathways is particularly important for ultra-rare conditions where commercial incentives are weakest.
The objective is not to replace market mechanisms but to design complementary structures that address their limitations.
Rare disease requires systems that operate effectively over extended timeframes under conditions of uncertainty and evolving knowledge.
This has implications for governance at multiple levels.
Strategic planning must adopt long-horizon perspectives, recognising that returns on investment may take years or decades to materialise. Risk management frameworks must accommodate uncertainty rather than attempt to eliminate it, incorporating scientific and operational variability into decision making.
Capacity building requires sustained investment in workforce, infrastructure, and research. This includes training clinicians, developing data systems, and supporting patient organisations.
Learning systems are essential. Rare disease systems must be designed to continuously capture data, evaluate outcomes, and adapt policies and practices accordingly.
Long-horizon design is a practical requirement for system resilience.
At its core, the rare disease challenge is one of alignment.
Alignment between policy and practice. Alignment between data systems and clinical needs. Alignment between incentives and outcomes. Alignment between short-term actions and long-term goals.
Governance provides the structure through which this alignment can be achieved.
Effective governance in this context requires clarity of roles and responsibilities across stakeholders. It requires transparency in decision-making and prioritisation. It requires integration across sectors and disciplines. It requires a sustained focus on long-term outcomes rather than short-term metrics.
This is not a one-time intervention. It is an ongoing process of system design and refinement.
Rare disease exposes the limitations of systems designed for scale, predictability, and short-term efficiency. It requires a different approach that prioritises coordination, data integration, and long-horizon thinking.
Framing rare disease as a systems challenge shifts the focus from isolated interventions to structural design. It highlights the role of governance in creating conditions where policy, data, and care can operate coherently.
The question is not whether solutions exist for individual components. Many do. The question is whether systems are designed to integrate these components effectively over time.
In rare disease, the answer to that question determines whether progress remains incremental or becomes transformative.