In rare disease research, one of the most important and most frequently misunderstood steps is proving that a specific gene actually causes a specific disease. Modern sequencing technologies make it relatively easy to identify genetic variants, but identifying a variant is not the same as proving causation. Every individual carries thousands of genetic differences compared with the reference genome, and the vast majority of these differences have no effect on health.
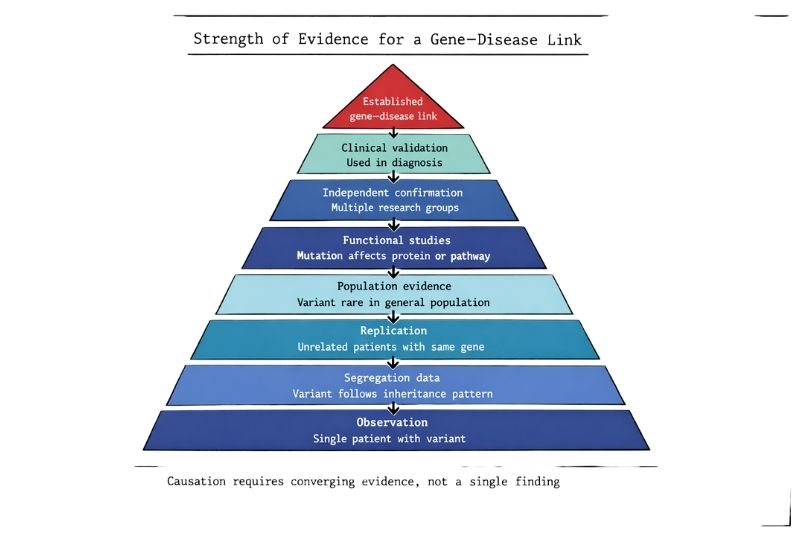
For this reason, the central challenge in rare disease genetics is not finding variants but determining which variant is responsible for the clinical condition. This requires a structured and rigorous methodology. Scientific credibility depends on demonstrating that the relationship between a gene and a disease is reproducible, biologically plausible, and supported by multiple independent lines of evidence.
Because rare diseases involve small patient numbers, the process of confirmation is often slower and more complex than in common diseases. However, the same standards must apply. A gene–disease link cannot be accepted on the basis of a single observation. It must be built step by step through clinical, genetic, experimental, and statistical evidence.
Understanding this process is essential for researchers, clinicians, patients, and policymakers, because it explains why rare disease discovery takes time and why methodological rigour is necessary to maintain trust in the field.
When sequencing identifies a mutation in a patient, the first question is whether the mutation is related to the disease at all. Humans naturally carry millions of genetic variants. Many are rare, and some appear unusual, but rarity alone does not mean pathogenicity.
An association means that a variant is found in a person with a disease.
Causation means that the variant produces the disease.
Establishing causation requires evidence from several independent sources. A variant must be shown to occur in affected individuals, to be rare in unaffected populations, to affect gene function, and to fit with the known biology of the condition. Without this combination of evidence, the finding remains a candidate rather than a confirmed cause.
Rare disease research must be particularly cautious at this stage, because small numbers increase the risk of coincidence.
The starting point for most gene discovery studies is careful clinical observation. Researchers must first define the phenotype precisely before searching for a genetic cause.
Phenotype definition includes
• Detailed clinical history
• Physical examination
• Laboratory investigations
• Imaging studies
• Age of onset
• Pattern of progression
• Family history
Two patients may appear similar but have different conditions. Conversely, the same genetic disorder may present differently in different individuals. Without accurate phenotyping, genetic results cannot be interpreted reliably.
In rare diseases, phenotype definition often requires collaboration between clinicians, geneticists, and specialist centres.
Once the phenotype is defined, sequencing is used to identify possible genetic causes. Common approaches include whole exome sequencing, whole genome sequencing, and targeted gene panels.
Sequencing typically identifies thousands of variants in each individual. Researchers therefore apply filtering strategies to narrow the list.
Typical filters include
• Variants rare in the general population
• Variants predicted to affect protein function
• Variants consistent with inheritance pattern
• Variants in biologically relevant genes
At this stage, the result is not proof. It is only a shortlist of candidates.
The next steps are required to determine which candidate, if any, is responsible for the disease.
One of the strongest forms of evidence comes from studying inheritance within families.
If a variant causes a disease, it should follow the expected inheritance pattern. For example, in a dominant disorder, affected individuals should carry the mutation, while unaffected relatives should not. In recessive disorders, affected individuals should carry two copies, while carriers have one.
Segregation analysis can confirm that the genetic change tracks with the disease across generations.
However, many rare diseases occur sporadically or involve small families, which limits the power of this method. When family data are limited, additional evidence becomes essential.
Confidence increases when unrelated patients with similar clinical features are found to have mutations in the same gene.
Because rare diseases are uncommon, these cases are often identified through international collaboration. Researchers use global databases, gene matching platforms, and research networks to find similar cases.
Tools such as gene matching services allow investigators in different countries to connect when they are studying the same gene.
Replication in independent patients reduces the likelihood that the finding is coincidental. In rare disease research, this step may take years because patients are widely distributed.
A key requirement for proving pathogenicity is showing that the variant is rare in the general population.
Large genomic databases contain sequence data from tens or hundreds of thousands of individuals. These databases allow researchers to determine whether a variant occurs in healthy people.
If a variant is common in the general population, it is unlikely to cause a severe rare disease. Population frequency data therefore provide an essential control.
Databases also help identify whether different variants in the same gene occur in unrelated patients, which strengthens the case for causation.
Genetic data alone are often not sufficient. Researchers must show that the mutation changes the function of the gene or protein.
Functional studies may involve
• Cell culture experiments
• Protein activity measurements
• Gene expression analysis
• Animal models
• Gene editing techniques
• Biochemical assays
For example, if a mutation affects an enzyme, researchers may test whether enzyme activity is reduced. If a gene is involved in development, animal models may be used to see whether the mutation produces similar abnormalities.
Functional evidence provides biological plausibility. Without it, the link between gene and disease may remain uncertain.
A proposed gene–disease relationship must also make sense in the context of existing knowledge.
Researchers examine whether the gene is expressed in the affected tissues, whether it participates in known biological pathways, and whether related genes cause similar disorders.
For example, a mutation in a nerve-specific protein is more plausible in a neurological condition than in a skin disorder.
Consistency with established biology does not prove causation, but it strengthens the argument and guides further experiments.
To maintain consistency across studies, genetic variants are classified using internationally accepted criteria.
Variants are typically assigned to categories such as pathogenic, likely pathogenic, uncertain significance, likely benign, or benign.
Classification depends on multiple factors, including
Using standardised guidelines prevents premature claims and ensures that different laboratories apply the same criteria.
This step is essential for maintaining scientific credibility, particularly in rare disease research where data are limited.
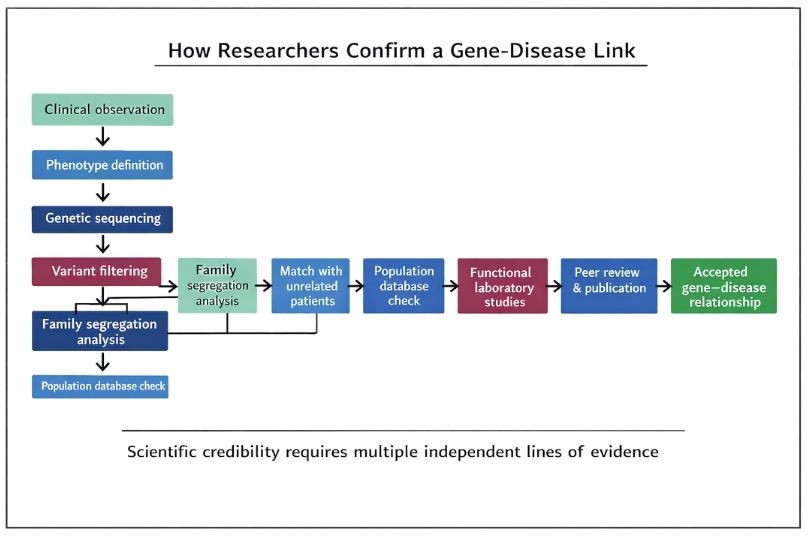
A gene–disease link is not considered established until the findings have been reviewed by other scientists and published in a peer-reviewed journal.
Peer review evaluates study design, data quality, statistical methods, and interpretation. Reviewers may request additional experiments or clarification before publication.
Although peer review is not perfect, it provides an important level of independent scrutiny.
Publication also allows other researchers to examine the evidence and attempt replication.
True confirmation requires independent replication by other groups.
Replication may involve finding additional patients with mutations in the same gene, demonstrating similar functional effects in different laboratories, or confirming the clinical features in new populations.
Replication reduces the risk of bias, technical error, or coincidence.
In rare disease research, replication may take many years because of the small number of cases worldwide. However, without replication, confidence remains limited.
Scientific evidence alone is not enough. A gene–disease link must also be validated for clinical use.
Clinical validation includes confirmation in accredited laboratories, use in diagnostic testing, and inclusion in clinical guidelines.
Diagnostic laboratories follow strict standards before reporting a variant as disease-causing. This protects patients from incorrect diagnoses and inappropriate treatment decisions.
Clinical validation ensures that research findings can be applied safely in healthcare.
When evidence is strong, the gene–disease link may be included in clinical databases, diagnostic panels, and medical guidelines.
At this stage, the relationship is considered established, although it can still be revised if new evidence appears.
Science remains open to correction, and reclassification of variants does occur as knowledge improves.
Rare disease research faces unique challenges.
Patient numbers are small, families may be scattered across countries, funding is limited, and functional studies can be difficult to perform. Some genes have subtle effects that are hard to detect, while others affect biological pathways that are not yet fully understood.
Because of these challenges, confirmation often requires international collaboration and long-term data collection.
The time required is not a sign of inefficiency. It reflects the need for reliable evidence.
Declaring a gene–disease link too early can have serious consequences.
Patients may receive incorrect diagnoses. Families may be given inaccurate genetic counselling. Research funding may be directed toward the wrong target. Trust in science may be damaged.
For these reasons, researchers are cautious before claiming causation. Scientific credibility depends not only on discovery but also on restraint.
Rare disease research depends heavily on cooperation between centres.
International registries, shared databases, and collaborative networks allow small numbers of cases to be combined. Without this cooperation, many rare disease genes would never be identified.
Collaboration also improves data quality and reduces the risk of false conclusions.
Patient registries provide essential information for confirming gene–disease links.
Registries collect clinical data, genetic results, and long-term outcomes. They allow researchers to identify similar cases and to study the natural history of the disease.
For rare conditions, registries may be the only way to gather enough evidence to establish causation.
Modern sequencing and bioinformatics have made gene discovery faster, but confirmation still requires the same careful methodology.
New technologies allow researchers to identify candidate genes quickly, but functional testing, replication, and clinical validation still take time.
Technology increases speed, but it does not replace scientific rigour.
Scientific credibility depends on transparency, reproducibility, and independent verification.
Rare disease communities often face urgent needs, but urgency cannot replace evidence. Reliable diagnoses and effective treatments depend on careful methodology.
A gene–disease link must be supported by clinical observation, genetic data, functional studies, replication, and peer review before it can be accepted.
Confirming a link between a gene and a disease is a complex process that requires multiple independent lines of evidence. In rare diseases, the process is particularly challenging because patient numbers are small and data are limited.
Strict methodology is essential. Careful phenotyping, rigorous genetic analysis, functional testing, replication, and clinical validation all contribute to scientific credibility.
The time required to establish a gene–disease relationship is not a weakness of the system. It is a necessary safeguard that protects patients, strengthens research, and ensures that rare disease discoveries are reliable.
Understanding this process helps explain why progress can appear slow, and why that slow progress is often the result of careful science rather than a lack of effort.