Developing treatments for rare diseases presents unique challenges that extend far beyond the complexities faced in mainstream drug development. While patients with common conditions often have multiple treatment options available within years of initial research, those living with rare diseases may have to wait for decades for their first therapeutic option and that too if one ever becomes available at all.
Currently, fewer than 10% of rare diseases have FDA-approved treatments, leaving millions of patients without specific therapeutic options. Understanding why these treatments take longer to reach patients requires examining the various interconnected barriers that include scientific research, economic viability, regulatory approval, and healthcare delivery systems.
The fundamental obstacle in the development of drugs for rare diseases lies in the mathematics and process of clinical trials. Traditional pharmaceutical research relies on large patient populations to generate statistically meaningful data, but rare diseases affect small numbers of people by definition.
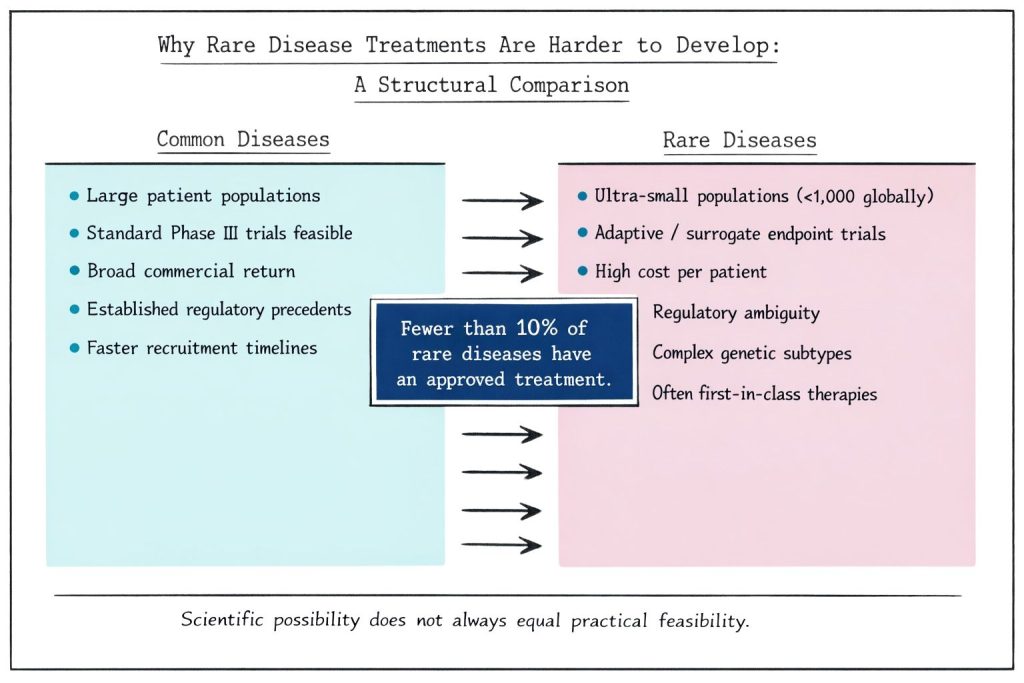
For ultra-rare conditions affecting fewer than 1,000 patients globally, conducting conventional Phase III clinical trials becomes virtually impossible. Researchers often struggle to recruit sufficient participants to achieve significant statistical power, particularly when patients are dispersed across different countries and continents. This scarcity forces pharmaceutical companies to develop alternative research methodologies and seek specialised regulatory pathways.
The regulatory landscape has adapted to these constraints through accelerated approval mechanisms that accept surrogate endpoints rather than traditional clinical outcomes. However, these alternative pathways still require substantial evidence, and generating that evidence with limited patient populations remains a challenge in terms of resources and time.
Gene therapy development exemplifies these challenges. For conditions like Duchenne muscular dystrophy, where multiple genetic mutations exist within the same gene, researchers must develop separate animal models for each specific mutation. This preclinical work alone can extend development timelines by 2+ years before human trials even begin.
The economics of rare disease drug development create additional hurdles that delay the treatment availability. Pharmaceutical companies have to recover development costs, often exceeding $1 billion per approved drug, from much smaller patient populations compared to common diseases.
Research has identified a critical threshold known as the "frontier of feasibility," representing the minimum patient population size at which developing a therapy becomes economically viable. This economic reality explains why many rare diseases remain without treatment options despite existing scientific knowledge about potential therapeutic targets.
Several strategies can improve economic viability and expand this frontier of feasibility:
Beyond clinical trials, rare disease treatments, particularly advanced therapies such as gene and cell therapies, involve extensive preclinical development phases that significantly extend the overall timeline.
The regulatory submission process for gene therapies includes multiple sequential steps, like preparing regulatory documents (1-2 months), completing the pre-Investigational New Drug application process (approximately 3 months), compiling study reports and data (2 months), and awaiting FDA approval (30 days). Each phase requires meticulous attention to detail and often multiple iterations based on regulatory feedback.
For gene therapies using viral vectors, such as adeno-associated viruses (AAVs), additional complexities may arise from potential immune responses. Patients may require immunosuppressive medications before treatment administration, creating additional safety considerations and monitoring requirements that extend both development and implementation timelines.
The historical lack of clear regulatory guidance for rare disease treatments creates uncertainty for developers, often requiring extensive pre-submission meetings and iterative discussions with regulatory agencies. The FDA's recent Rare Disease Evidence Principles (RDEP) represent significant progress by clarifying that sponsors can use strong mechanistic evidence, biomarker data, non-clinical models, clinical pharmacodynamic evidence, and case reports, and not exclusively traditional pivotal trials.
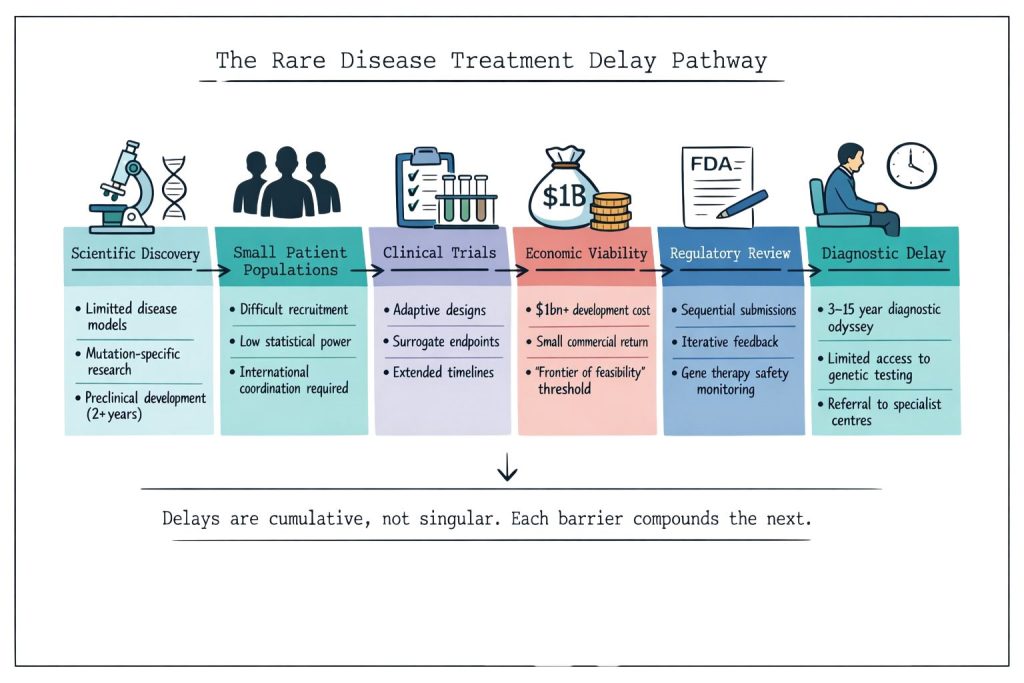
An often-overlooked barrier to treatment access is the extended diagnostic journey that rare disease patients experience. On average, patients spend 3-15 years receiving an accurate diagnosis, during which time they cannot access condition-specific treatments, even if they exist.
This diagnostic delay creates a practical barrier to treatment access that operates independently of drug development timelines. Patients may receive multiple misdiagnoses, undergo unnecessary procedures, and experience symptom progression while searching for answers. The complex, multi-system nature of many rare diseases requires specialised expertise that may not be readily available in all healthcare settings.
Genetic testing has improved diagnostic capabilities significantly, but access to comprehensive genetic testing remains inconsistent across healthcare systems. Some patients receive diagnoses only after treatments become available, creating additional delays in accessing potentially life-changing therapies.
Despite persistent challenges, the rare disease treatment landscape has evolved substantially in recent years. Over 1,430 clinical trials have been conducted worldwide for rare diseases since 2020, with neuromuscular and metabolic disorders leading research efforts.
Major pharmaceutical companies have significantly shifted their research portfolios toward rare diseases. By the end of 2024, two-thirds of drugs in some companies' pipelines targeted rare indications, representing a dramatic change from historical patterns focused on common conditions.
Several factors contribute to this increased attention:
Emerging technologies and methodologies are beginning to address traditional barriers in rare disease drug development. Platform technologies allow researchers to develop multiple treatments using similar approaches, reducing per-program development costs and timelines.
Natural history studies and patient registries provide valuable data about disease progression and potential treatment endpoints, supporting more efficient clinical trial design. These resources help researchers identify appropriate outcome measures and reduce the time needed to demonstrate treatment efficacy.
Collaborative research networks and international partnerships enable pooling of patient populations across geographic boundaries, making larger studies feasible for even ultra-rare conditions. These collaborations require significant coordination but can dramatically improve research efficiency.

The fundamental structural challenges, such as small populations, high development costs, and complex science, will continue to create longer development timelines for rare disease treatments compared to those for common diseases. However, continued innovation in research methodologies, regulatory frameworks, and collaborative approaches offers hope for accelerating the path from laboratory discovery to patient access.
Understanding these barriers helps patients, families, and healthcare providers maintain realistic expectations while supporting continued advocacy for improved research funding, regulatory efficiency, and international collaboration in rare disease research. Every incremental improvement in these systems brings treatments closer to the patients who need them most.