Rare disease research operates within a complex ethical area that differs significantly from conventional clinical research. With approximately 7,000 known rare diseases affecting 300 million people worldwide, researchers face and navigate unique challenges while protecting vulnerable populations and advancing scientific knowledge.
The rarity of these conditions creates inherent ethical dilemmas. Small patient populations, limited funding, and urgent medical needs combine to create situations in which traditional research ethics frameworks do not work and require careful adaptation. Unlike common diseases, where large patient pools allow for standard clinical trial designs, rare disease research demands innovative approaches that balance scientific rigour with patient welfare.
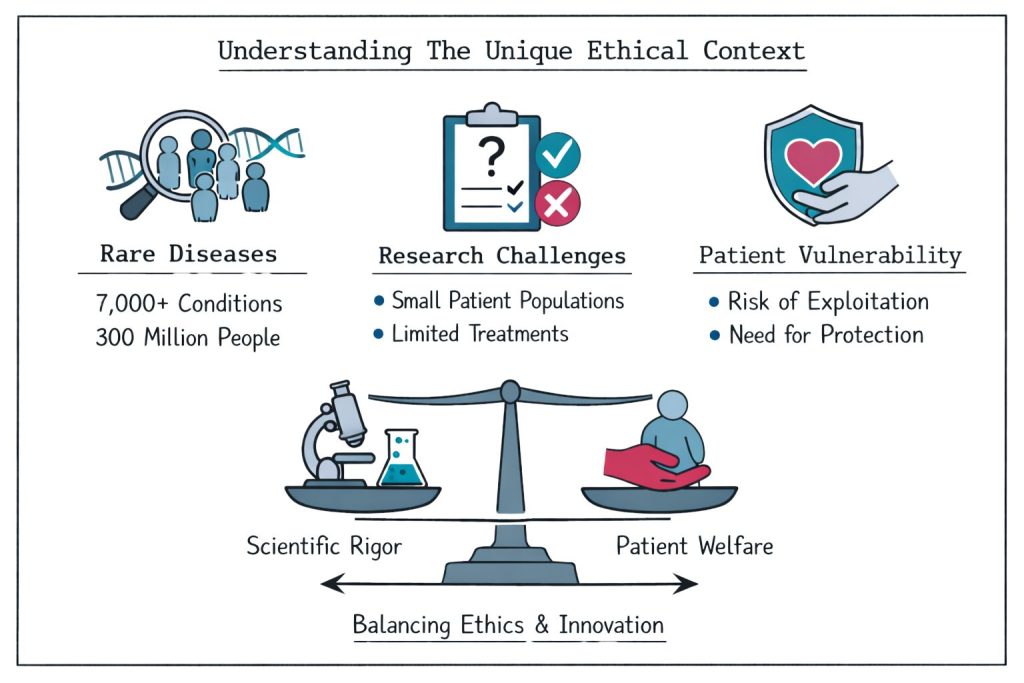
The vulnerability of rare disease patients adds another layer of complexity. Many individuals face limited treatment options, making them potentially susceptible to exploitation in research settings. This vulnerability necessitates heightened ethical scrutiny and protective measures.
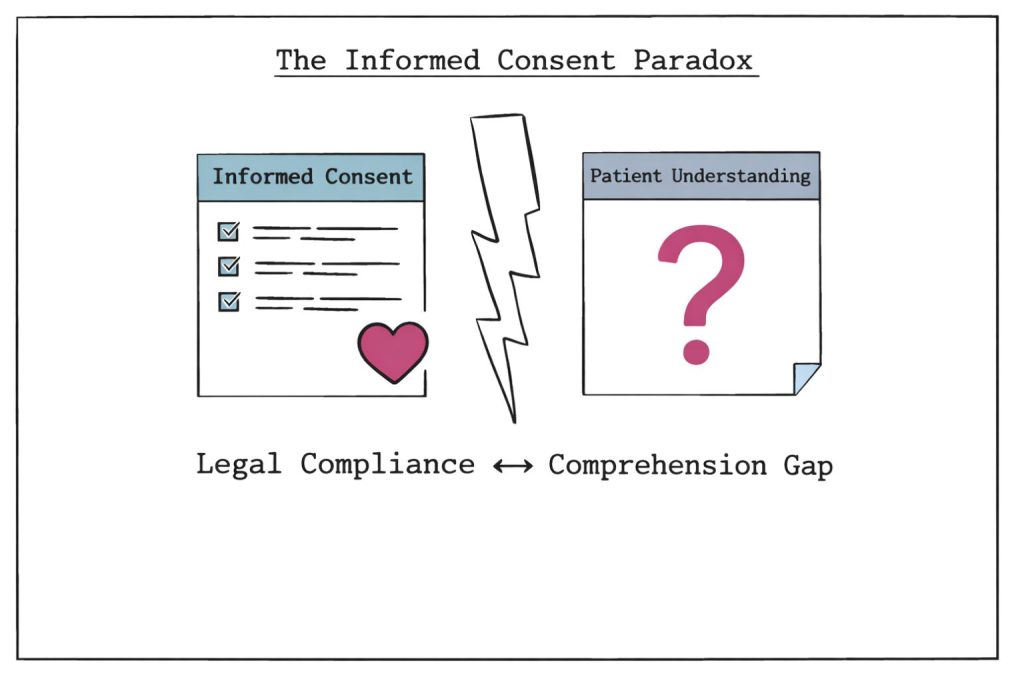
Traditional informed consent processes present significant challenges in rare disease research. Standard consent forms often contain lengthy, technical documents that prioritise legal protection over genuine patient understanding. This creates what researchers term the "informed consent paradox", which is the disconnect between legal compliance and meaningful comprehension.
The challenge intensifies when considering varying literacy levels and cultural differences across patient populations. Healthcare workers frequently lack sufficient knowledge about rare diseases, which can compromise the quality of information provided during the consent process. This knowledge gap may contribute to problematic attitudes toward patients and complicate recruitment efforts.
Rare disease populations are typically small and geographically dispersed, requiring creative recruitment strategies. Face-to-face recruitment often yields favourable patient responses but demands substantial time and financial resources. Researchers must balance the need for comprehensive patient enrollment with practical limitations and ethical obligations to minimise participant burden.
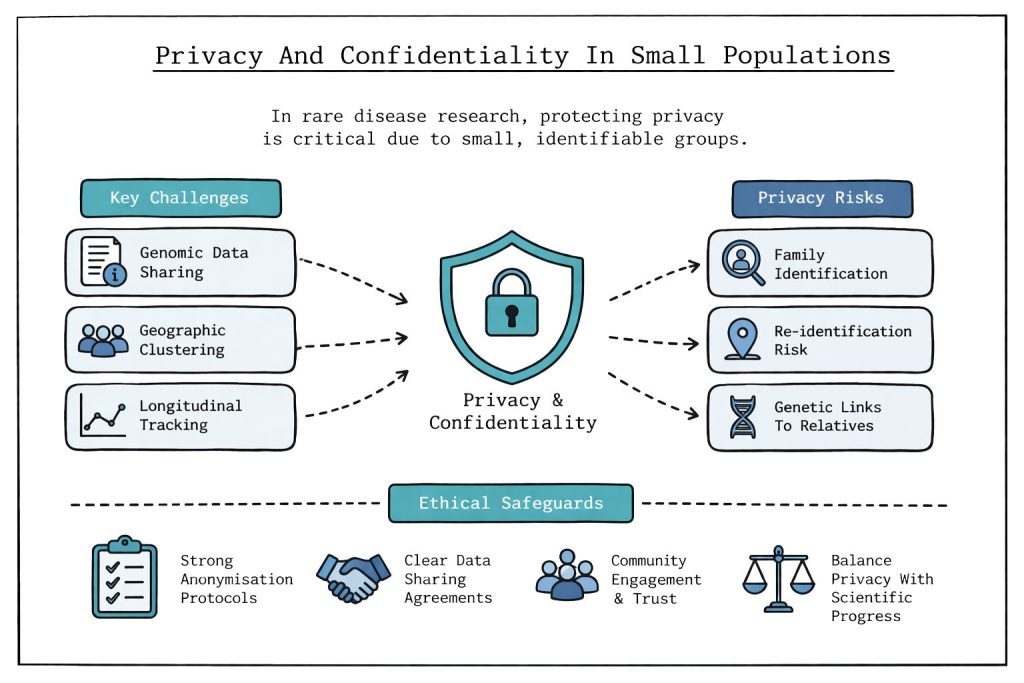
The protection of patient privacy becomes increasingly difficult as study populations shrink. In rare disease research, the smaller the patient cohort, the easier it becomes to identify individual participants within datasets, even when deliberate anonymisation efforts are implemented.
This creates a paradoxical situation in which rare disease patients exist within contained research environments, where true anonymity becomes nearly impossible to guarantee. The challenge extends beyond simple data protection to encompass:
Healthcare practitioners and patients must navigate these privacy challenges carefully, particularly when research involves genomic sequencing or stored biological samples that may have future research applications.
Institutional Review Board (IRB) oversight of rare disease research encounters significant structural challenges. Regulatory disparities exist between federally funded research covered by the Common Rule and FDA-regulated studies, and between federally funded research and non-federally funded research projects, creating inconsistent ethical standards across similar research activities.
Multi-site and multinational trials, often necessary for rare disease research, have expanded IRB responsibilities to address emerging research elements, including:
Research collaborations face significant obstacles when multiple IRBs require repeated reviews without substantial changes to study parameters. This redundancy slows scientific progress while consuming valuable resources that could otherwise support patient care or research advancement.
The 2015 Notice of Proposed Rule Making (NPRM) introduced eight proposed changes to the Common Rule aimed at streamlining approval processes while maintaining ethical protections. These modifications recognise the unique challenges faced by rare disease research communities.
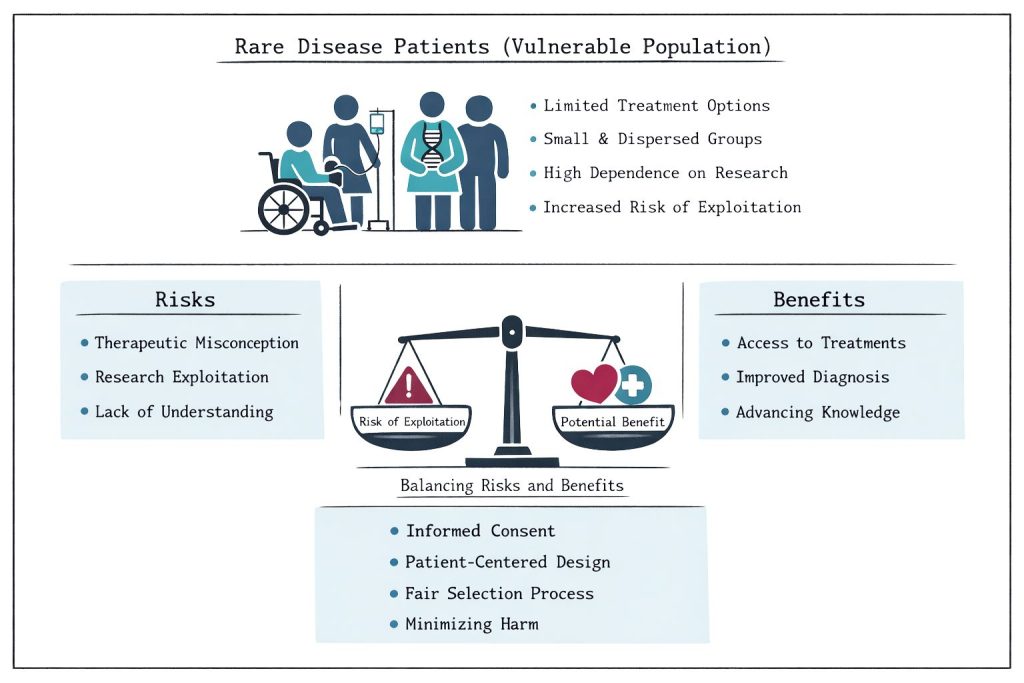
Rare disease patients represent an inherently vulnerable population, often lacking alternative treatment options. This vulnerability creates an ethical obligation to ensure research aligns with patients' genuine health priorities rather than exploiting their desperation for potential treatments.
According to the Declaration of Helsinki, trials involving vulnerable populations are ethically justified only when the research specifically addresses their health needs and offers realistic potential benefits. This principle requires careful consideration of:
Researchers must make ethically weighted decisions about enrollment criteria:
The inclusion of children in rare disease trials raises additional ethical considerations. Parents must make decisions on behalf of their children, but questions arise about whether certain trials should wait until children can make autonomous decisions about participation.
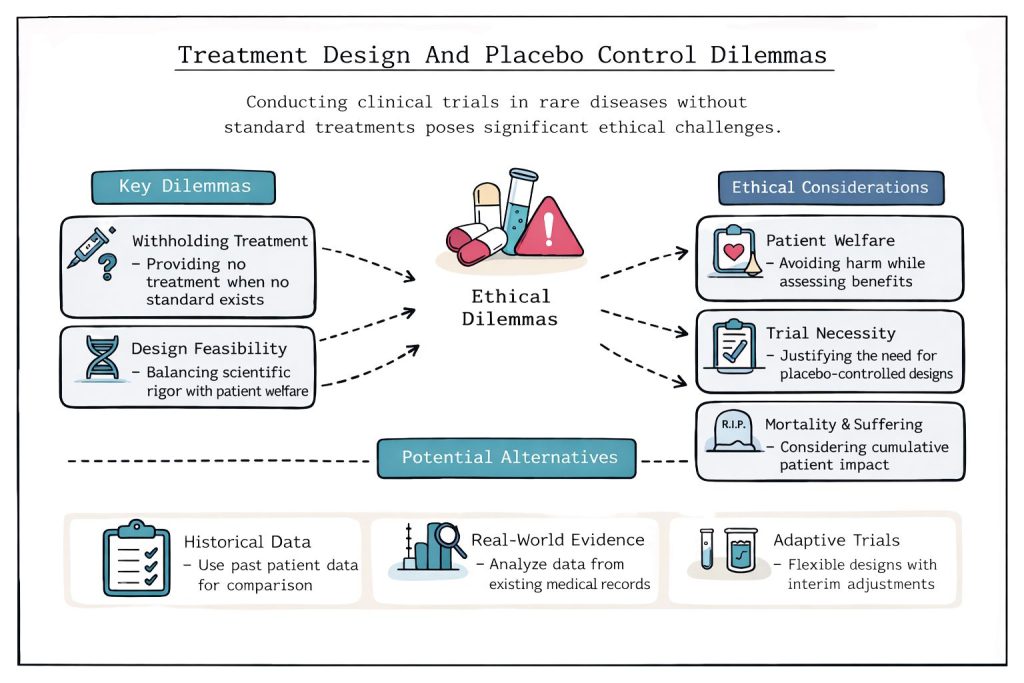
One of the most challenging ethical issues arises when conducting placebo-controlled trials in patients without available standard treatments. Researchers face the difficult decision of whether withholding potentially beneficial therapy is justified when no established alternatives exist.
Alternative study designs may provide ethical solutions:
These approaches raise methodological questions while potentially addressing ethical concerns about denying treatment to critically ill patients.
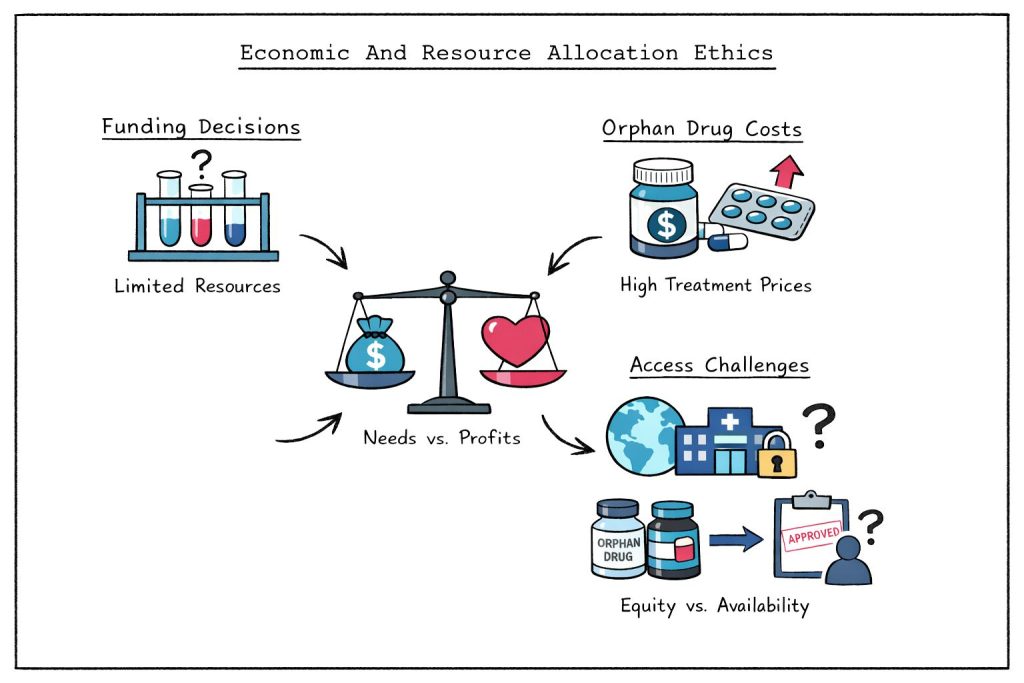
The decision to initiate rare disease research is complicated by economic realities, given that eventual markets for rare disease therapies may be quite limited. Funding organisations and pharmaceutical companies must balance economic feasibility with genuine health needs.
Some rare diseases attract numerous sponsors because they appear to offer straightforward drug-development pathways or to offer guaranteed financial returns, while most rare diseases receive no development attention whatsoever. This disparity raises questions about fair resource allocation across the rare disease community.
Patients with rare diseases often face complex decisions regarding their prognosis, limited treatment options, and substantial drug costs. Healthcare systems must grapple with questions regarding:
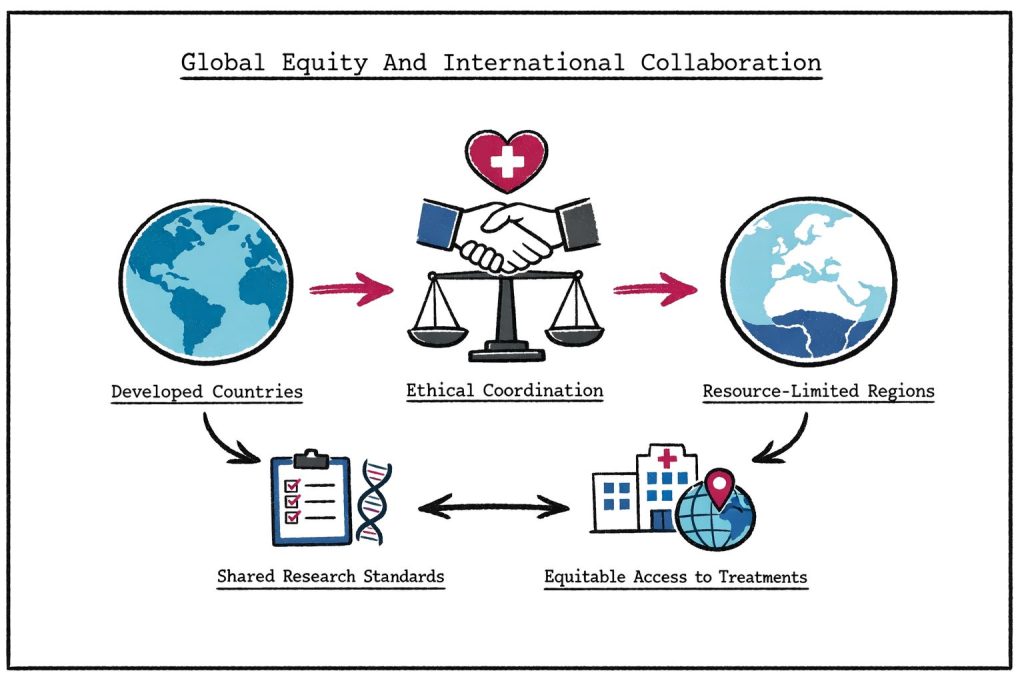
A significant ethical challenge involves conducting rare disease trials in resource-limited settings where approved therapies may not be immediately available due to cost or healthcare infrastructure limitations. While focusing exclusively on well-resourced regions simplifies trial logistics, this approach risks excluding valuable patient populations and perpetuating health disparities.
The ethical consideration extends beyond immediate access to therapy to encompass thoughtful, long-term planning that ensures future access pathways for study participants across diverse geographic regions.
International collaborations require coordination between different groups and countries, each potentially operating under different ethical frameworks and regulatory requirements. However, several questions persist about:
Genomic screening for rare diseases raises important ethical issues that extend beyond traditional clinical trial concerns. These emerging challenges include:
Currently, no participatory research projects have directly investigated ethical challenges experienced by patients with rare diseases or explored patient-centred solutions to these dilemmas. This represents an important gap in the research literature that requires attention from the research community.
Addressing the multifaceted ethical challenges in rare disease research requires sustained collaboration among regulators, researchers, patient communities, and advocacy groups. Success depends on fostering dialogue, maintaining transparency, and implementing patient-centred approaches that preserve ethical integrity while advancing life-changing therapies.
The complexity of rare disease research ethics demands continued attention and adaptive solutions. As research methodologies evolve and new technologies emerge, the ethical framework must evolve correspondingly to protect vulnerable populations while enabling scientific progress that serves their urgent medical needs.
Future developments in rare disease research ethics will likely focus on harmonising international standards, developing patient-centred outcome measures, and creating sustainable funding models that ensure equitable access to research opportunities and resulting treatments across diverse global populations.