Health policy plays a critical role in determining whether patients with rare diseases can access life-saving treatments. The regulatory roles and fundraising processes influence which therapies reach the market, how quickly they become available, and whether patients can afford them. Understanding these policy mechanisms reveals the challenges that affect patient access to care.
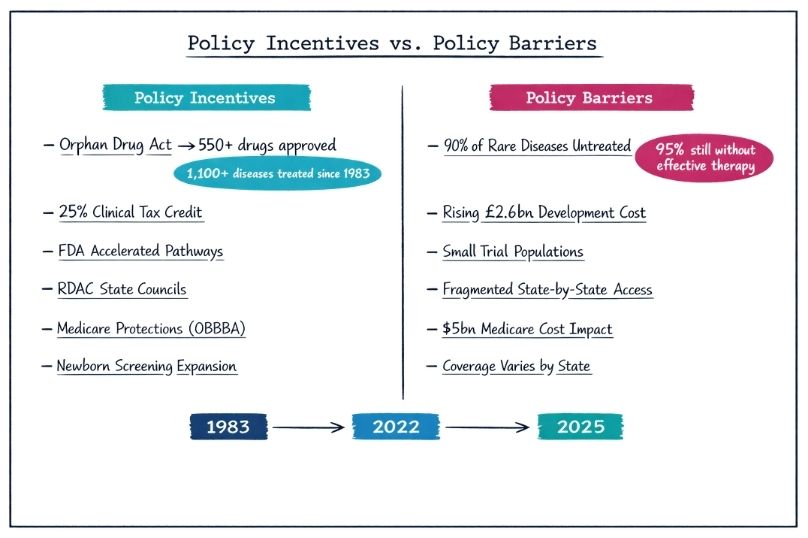
Before the Orphan Drug Act of 1983, only ten orphan drugs were approved in the preceding decade. The dramatic transformation following the Act’s implementation demonstrates the power of policy to reshape healthcare markets. Since then, more than 550 unique drugs and biologics have been approved for over 1,100 rare diseases. This progress is a direct result of targeted policy incentives, including tax credits, extended market exclusivity periods, and federal grant programs.
The Orphan Drug Tax Credit (ODTC) has been particularly effective in redirecting pharmaceutical investment toward previously neglected diseases. Economic analyses suggest that this incentive helped stabilise mortality rates from rare diseases while reducing the overall federal spending burden compared to earlier periods.
/
Following the Inflation Reduction Act of 2022, the policy environment underwent significant changes, granting Medicare the authority to negotiate drug prices. These changes had unintended consequences for rare disease treatments, prompting Congress to pass the One Big Beautiful Bill Act (OBBBA) in July 2025.
The OBBBA expanded protections for orphan drugs by excluding them from Medicare price negotiations if they are approved exclusively for rare diseases. While this measure was designed to preserve development incentives, it has raised concerns about potential costs to Medicare, estimated at $5 billion over ten years, as well as the risk of market manipulation if companies avoid expanding drug indications to maintain pricing advantages.
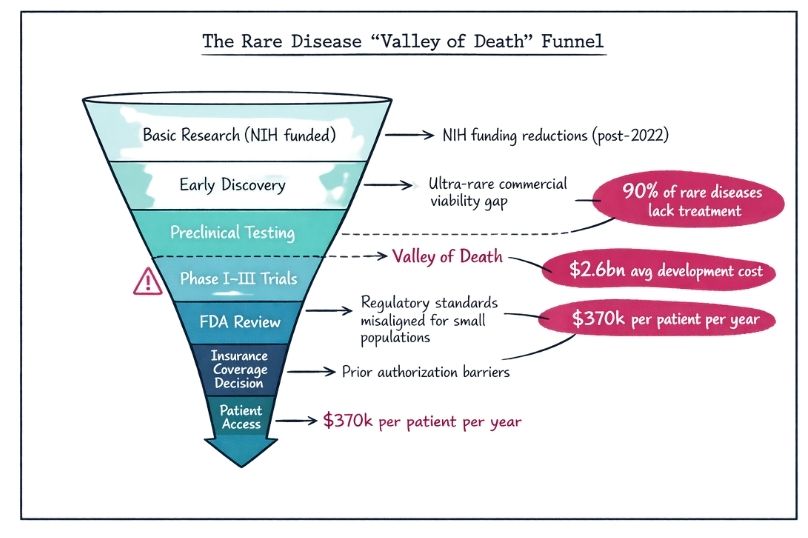
The National Institutes of Health (NIH) has historically been the backbone of rare disease research funding. However, policy decisions combined with rising inflation have significantly reduced NIH funding since 2022, leading to delayed or cancelled grants for rare disease research.
These funding reductions contribute to what researchers describe as the “valley of death” — a gap in which promising early-stage discoveries fail to progress to clinical trials. This challenge is particularly severe for ultra-rare conditions that lack clear commercial viability and affect the entire pipeline of potential treatments.
The FDA plays a central role in determining whether rare disease treatments become available through mechanisms such as accelerated approval pathways and specialised initiatives, including the Rare Disease Innovation Hub. These programs aim to support developers and attract investment in rare disease therapies.
However, regulatory requirements can also create barriers. Many approval standards were developed for common diseases and are difficult to meet in rare disease research due to small patient populations. Recent high-profile cases, including the rejection of a treatment for Barth syndrome despite expert support, highlight this misalignment.
Despite decades of policy incentives, more than 90% of rare diseases still lack approved treatments. The average cost of bringing an orphan drug to market has risen to approximately $2.6 billion, creating substantial economic barriers even with existing incentives.
Once approved, orphan drugs often cost more than $370,000 per patient per year, reflecting both development expenses and market dynamics shaped by policy frameworks. For the millions of individuals living without effective treatments, these figures represent more than statistics — they reflect a race against time.
Health policy shapes access to rare disease treatments through multiple economic mechanisms beyond regulatory approval. Federal funding priorities, tax incentives, and grant allocation decisions strongly influence which conditions receive research attention and which ultimately gain treatment options.
The Orphan Drug Tax Credit provides a 25% credit for qualified clinical testing expenses, significantly reducing financial risk for pharmaceutical companies. While this incentive encourages investment in otherwise unviable areas, any changes to its scope or value can directly affect industry investment decisions and future treatment availability.
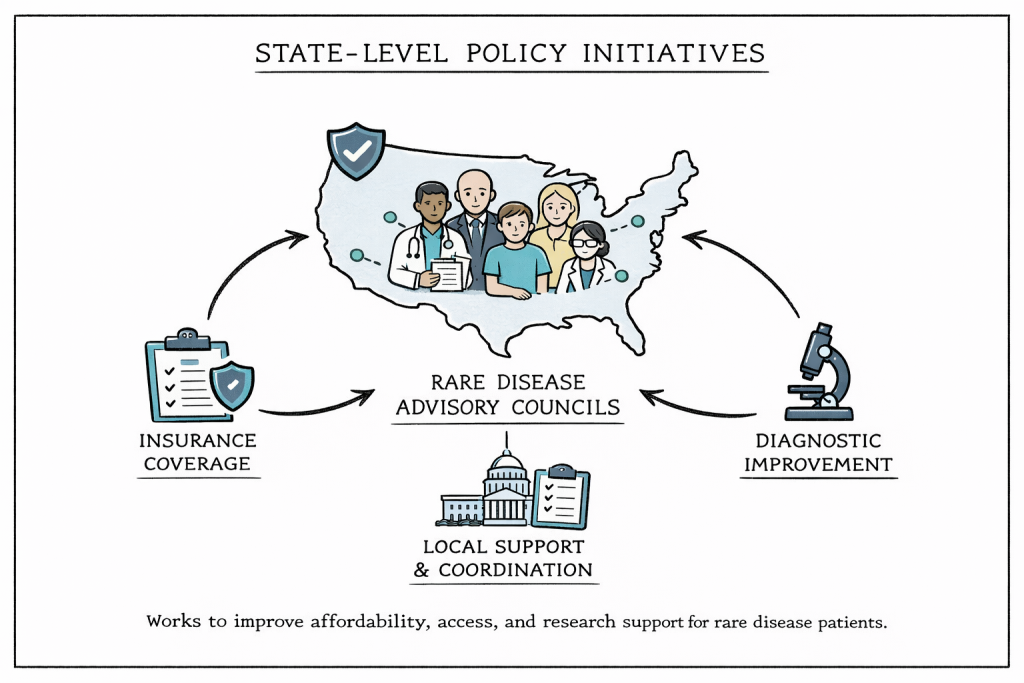
More than 20 U.S. states have established Rare Disease Advisory Councils (RDACs) that include patients, caregivers, healthcare professionals, insurers, researchers, and policymakers. These councils develop evidence-based recommendations to address local barriers related to affordability, access, and research support.
State-level policies often focus on insurance coverage mandates, improvements in diagnostic infrastructure, and coordination with federal programs. However, differences among state approaches create a fragmented system in which patient access depends heavily on geographic location, raising concerns about equity and consistency of care.
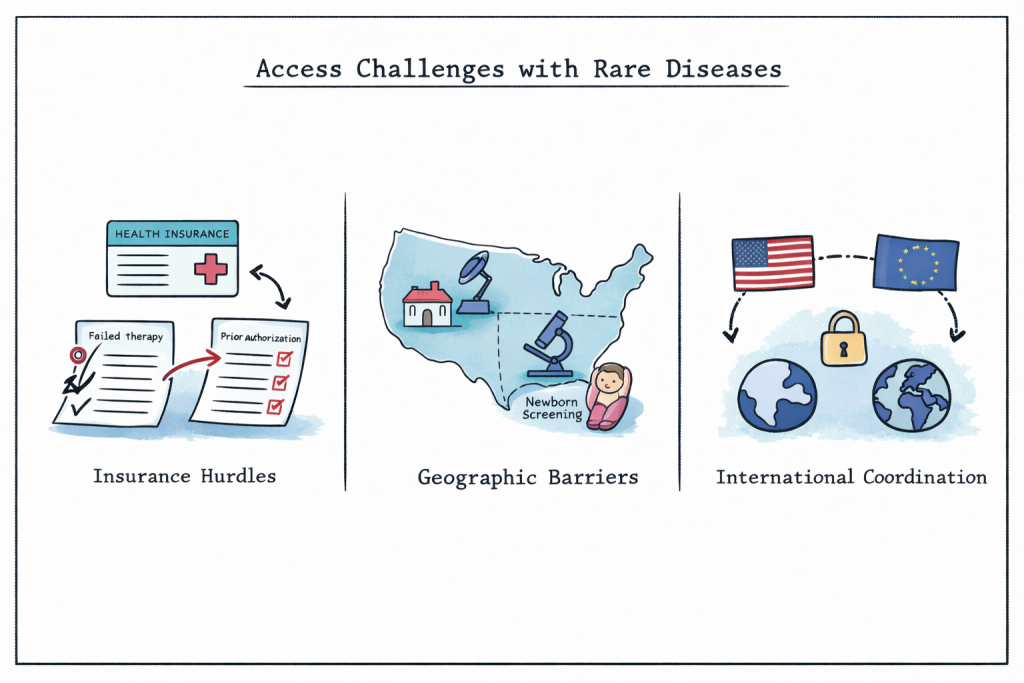
Even after treatments receive regulatory approval, patients frequently encounter significant access barriers. Insurance coverage decisions, formulary placement, and prior authorisation requirements can delay or restrict access to necessary therapies.
In some cases, prior authorisation protocols require patients to repeat previously unsuccessful treatments when changing insurance plans, delaying effective care by weeks or months. These delays can worsen health outcomes and increase overall healthcare costs due to complications or hospitalisation.
Geographic and diagnostic factors further limit access to rare disease care. Rural areas often lack experienced specialists, and diagnostic delays remain common nationwide. Federal initiatives that fund specialised centres and expand telemedicine services have begun to address these gaps and improve patient outcomes.
Newborn screening programs coordinated by the Centres for Disease Control and Prevention represent successful policy interventions that enable early diagnosis and timely treatment for certain rare conditions. However, screening coverage varies by state, resulting in unequal access to early intervention.
As rare disease research becomes increasingly global, international policy coordination plays a growing role in treatment access. Recent national security-driven restrictions on NIH funding have limited some international collaborations, potentially reducing access to the global patient populations needed for meaningful research.
European Union policies, particularly those guided by the European Medicines Agency’s orphan drug designation process, create parallel incentive structures that influence global development strategies. When regulatory and incentive frameworks are aligned across regions, treatments can reach patients more quickly. In contrast, policy divergence may lead to duplicated efforts, regulatory delays, and slower access to life-saving therapies.
Health insurance policies significantly influence whether patients can access rare disease treatments through coverage decisions, formulary placement, and prior authorisation requirements. Public programs such as Medicare and Medicaid directly affect vulnerable populations, while private insurers vary widely in their approaches to rare disease coverage.
The complexity of insurance systems often necessitates specialised patient advocacy and case management services. Policy interventions that standardise coverage processes and reduce administrative burden can meaningfully improve access, even without changes to treatment availability.
Experts increasingly emphasise the need for comprehensive policy approaches that address multiple barriers simultaneously. Proposed solutions include standardised care pathways, expanded funding for diagnostic infrastructure, and continued regulatory flexibility for ultra-rare conditions.
A central challenge remains balancing incentives for pharmaceutical innovation with cost-containment measures and patient access requirements. This balance is critical, given that approximately 95% of rare diseases still lack effective treatments.
New policies increasingly rely on collaboration among all stakeholders in the rare disease community. These coordinated efforts involve patients, researchers, industry representatives, regulators, and payers working together to develop comprehensive solutions that address the full spectrum of access barriers.
Coordination among federal agencies, state governments, and international bodies significantly impacts treatment availability. The future success of rare disease patients depends largely on the authorities’ ability to support innovation while ensuring standardised access to treatments.
Health policies will ultimately determine whether advances in rare disease research are transformed into accessible therapies for patients worldwide. Decisions regarding drug pricing, research funding allocation, and regulatory standards establish the framework within which future generations of rare disease patients will either gain access to life-saving treatments or continue to face barriers despite technological progress. In this context, effective funding sources for rare disease research are crucial for fostering innovation and bringing forward new therapies. Collaborative efforts among governments, private institutions, and non-profit organizations can enhance the landscape of available resources, ensuring that research initiatives receive the necessary support. By investing in these pathways, we can hope to pave the way for breakthroughs that meet the urgent needs of patients suffering from these often-overlooked conditions.
Rare Disease Day reminds us that behind every policy debate and pricing decision are real people waiting for treatment. Millions of patients worldwide still face delayed or denied access to appropriate therapies, not because the science failed them, but systems meant to support them often fall short.