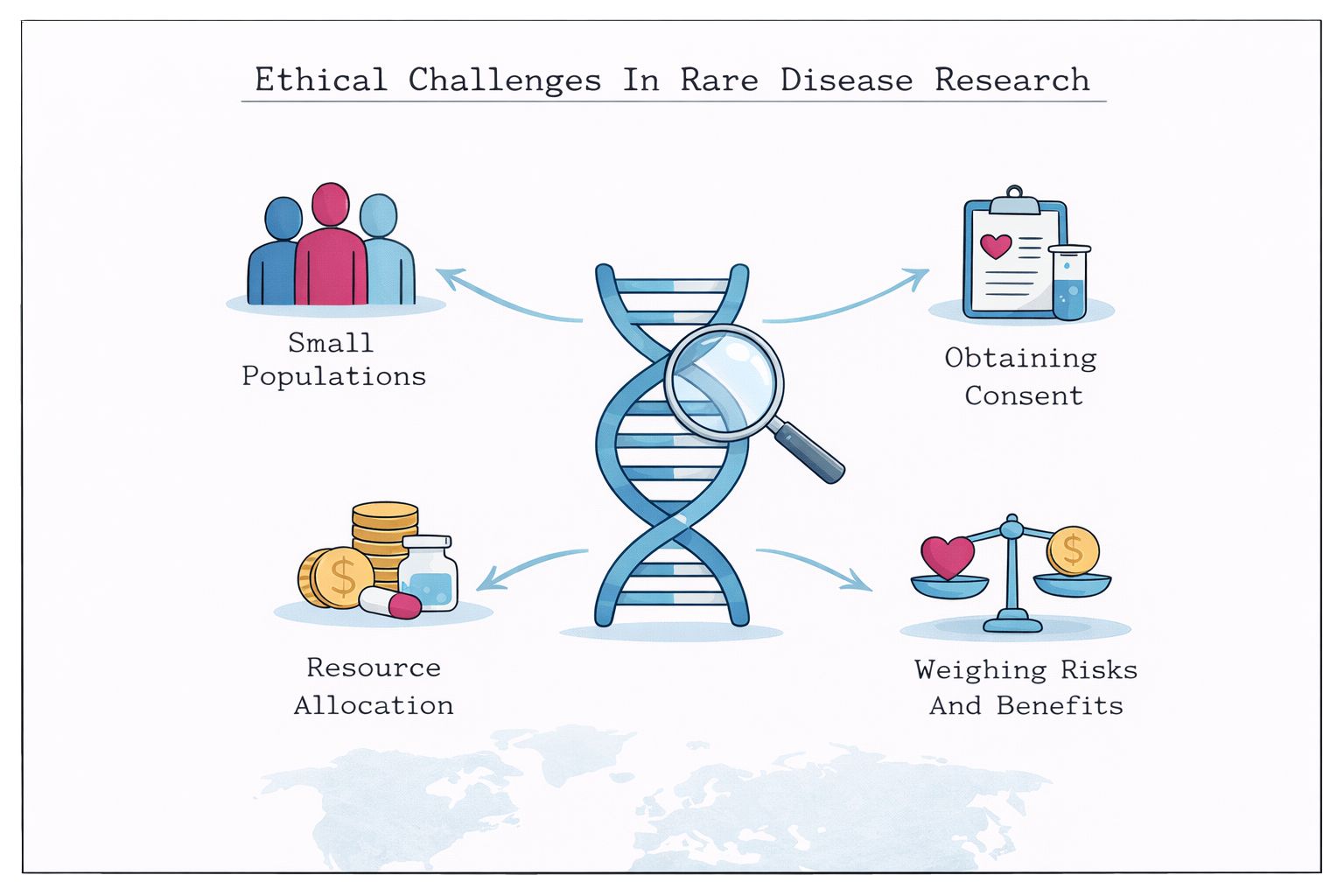
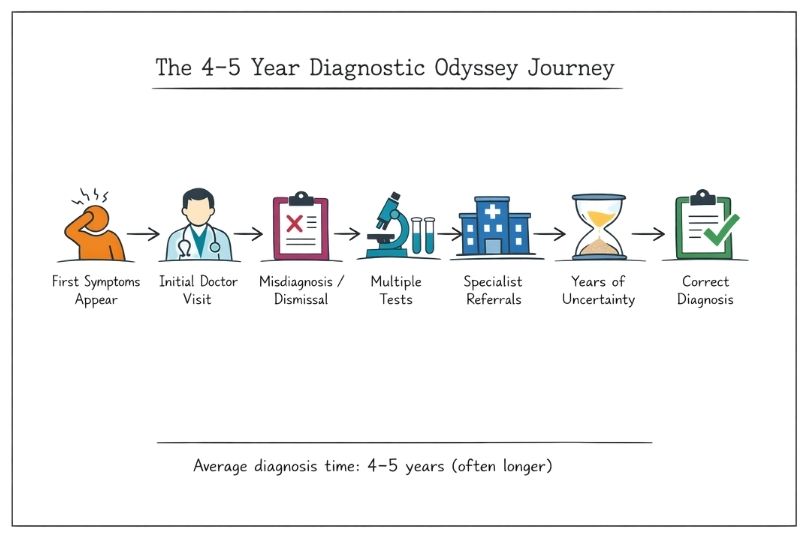
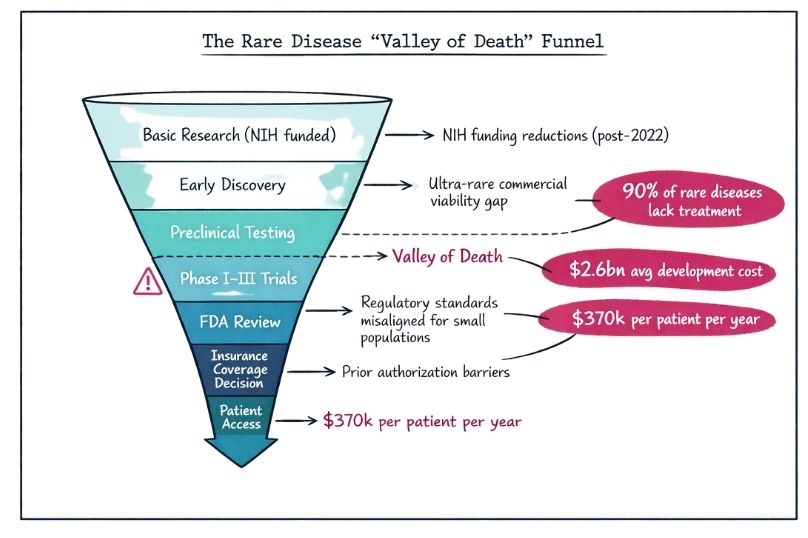
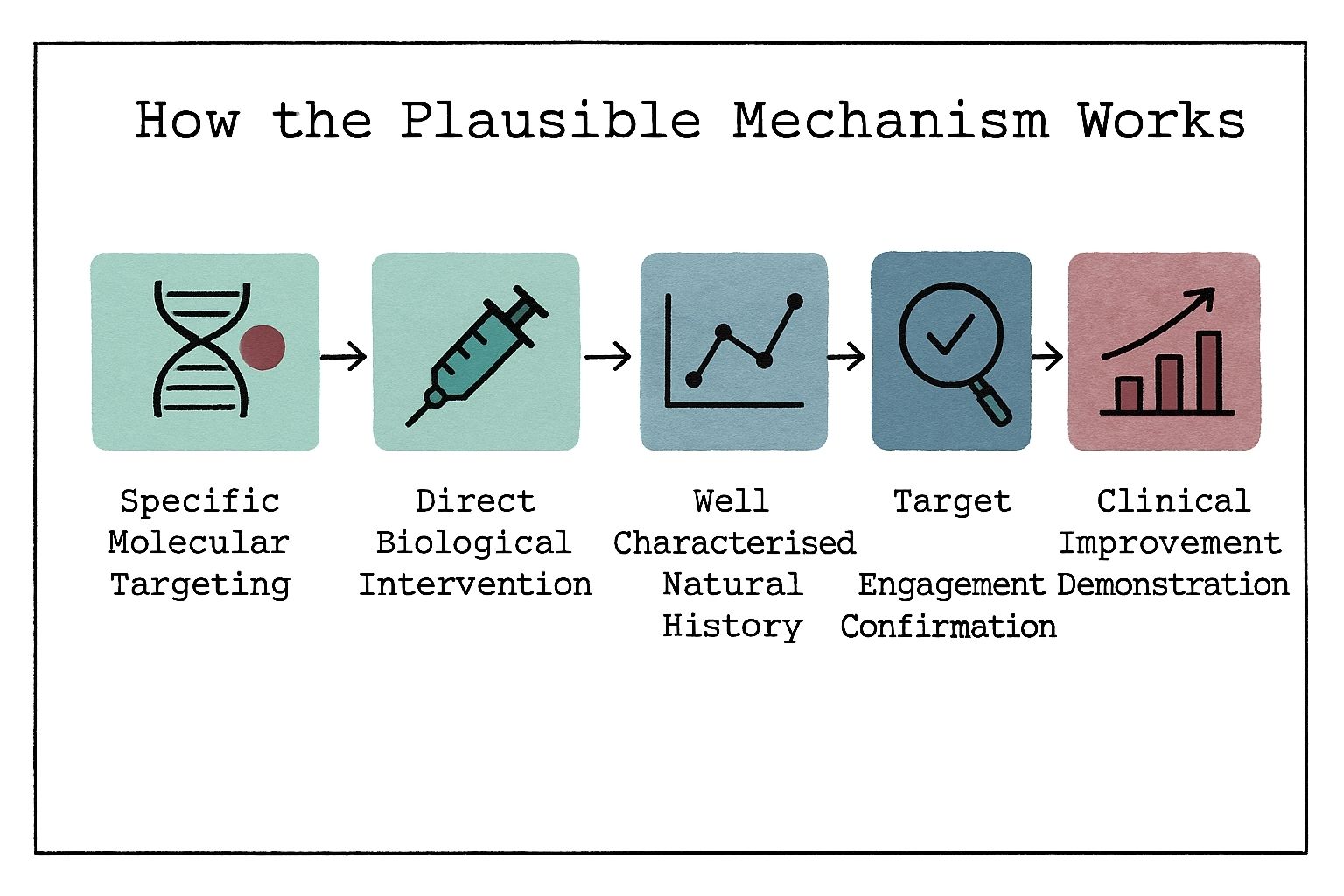
Learn how rare disease research challenges traditional ethics, including issues around patient recruitment, vulnerability, and regulatory oversight.
Decisions made by regulators, governments, and health authorities have a major impact on rare disease research, diagnosis, and access to treatment. This section explains the policies and regulatory frameworks that shape how medicines are approved, how clinical trials are run, and how healthcare systems decide which treatments are available to patients.
Articles cover topics such as orphan drug designation, approval pathways, health technology assessment, reimbursement decisions, international guidelines, and changes in national or global health policy.
We also report on recent regulatory decisions, consultations, and legislative updates that may affect patients, researchers, charities, and industry. The aim is to provide clear, evidence-based explanations of complex rules and processes, so readers can understand how policy and regulation influence real-world outcomes in rare disease.