Explore how endpoints are defined in rare disease clinical trials, the challenges of small populations, and the growing role of patient-reported outcomes in regulatory decisions.
Clinical trials are an essential part of rare disease research, but the process can be complex, technical, and difficult to navigate for patients and families.
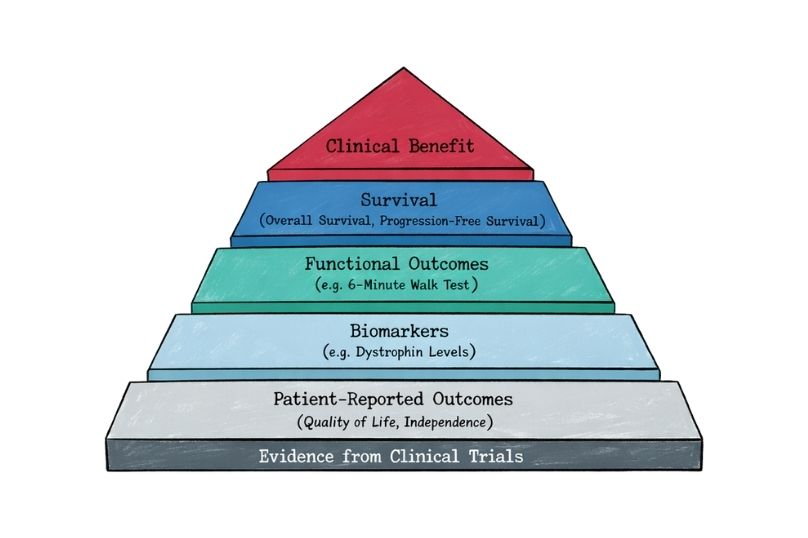
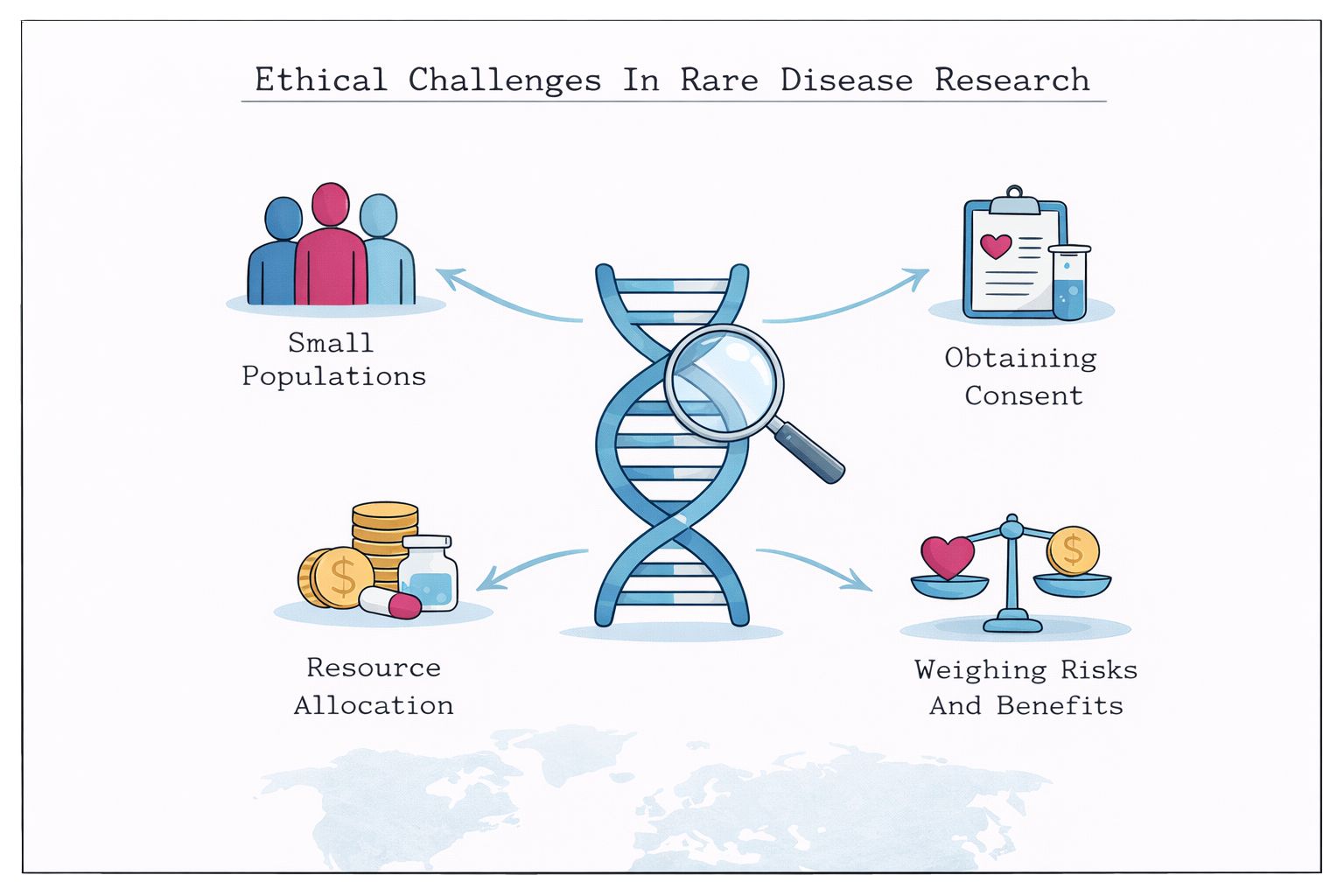
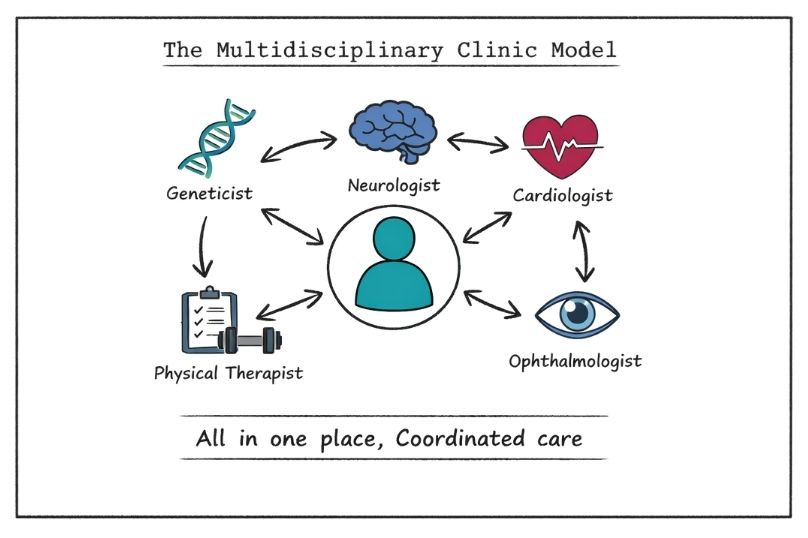
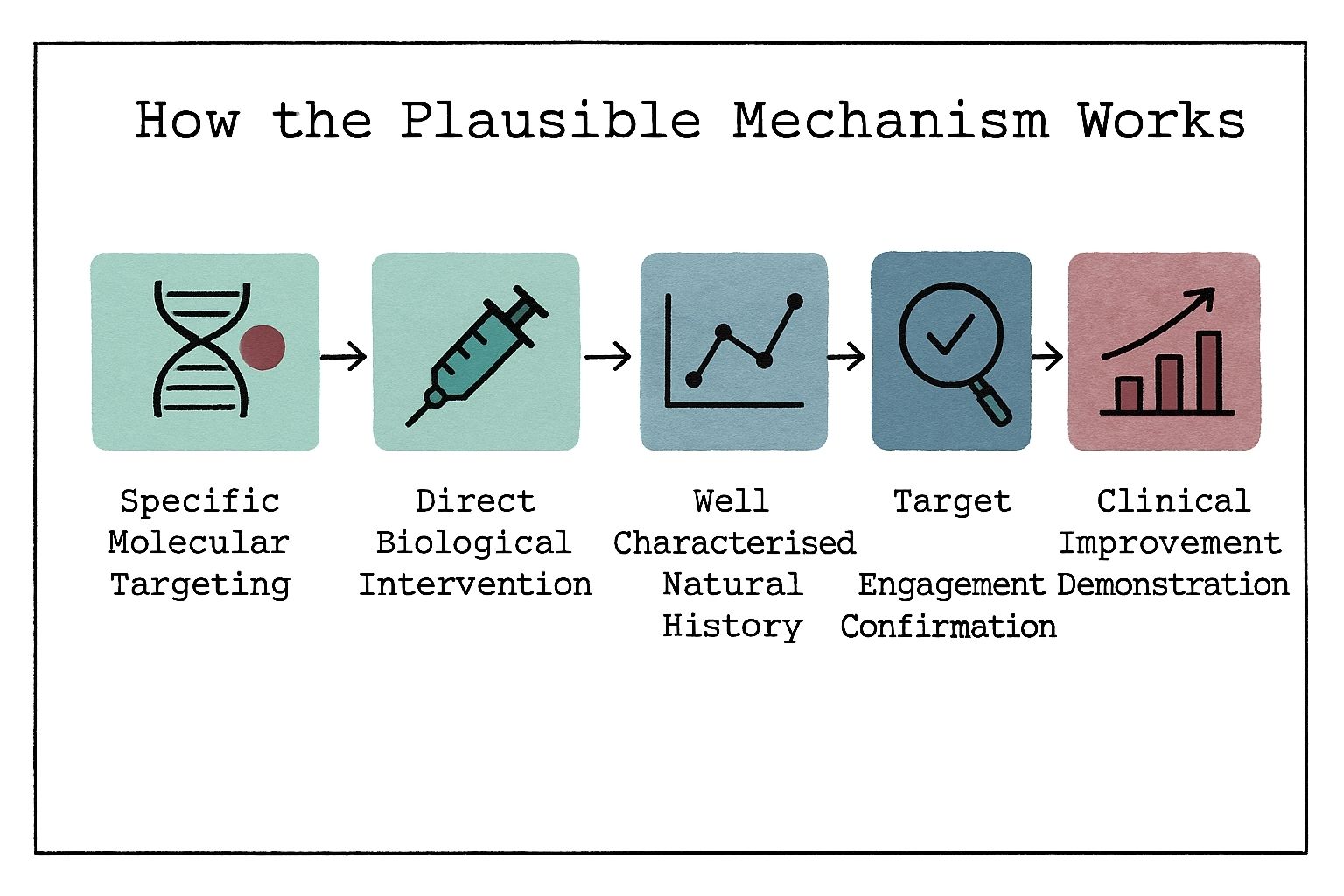
This section of Rare Disease Watch explains how clinical trials work, what different trial phases mean, how participants are selected, and what the risks and potential benefits may be.
Alongside practical guides, this section also covers recent clinical trial news, research announcements, regulatory decisions, and updates from pharmaceutical and biotechnology companies, helping readers stay informed about developments that may affect current or future treatment options.
Articles provide clear, evidence-based guidance on topics such as informed consent, eligibility criteria, placebo use, safety monitoring, regulatory approval, and what to expect before, during, and after a trial. The aim is to present accurate, balanced information so that decisions about participation can be made with realistic expectations and a clear understanding of the science.