Rare disease research operates within a uniquely complex funding ecosystem that brings together government agencies, patient organizations, academic institutions, and private foundations. Understanding how these funding ecosystems work helps explain why research progress varies so widely across the 7,000+ known rare diseases and highlights the strategic decisions that shape treatment development.
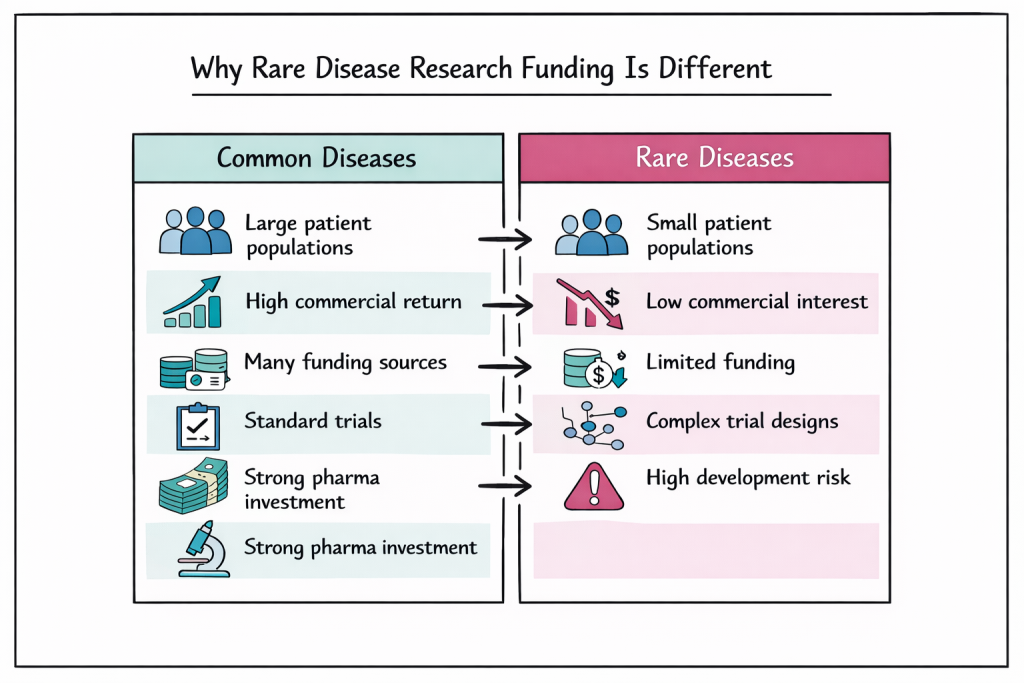
The funding landscape for rare diseases differs significantly from common condition research like diabetes or heart disease due to smaller patient populations, limited commercial interest, and higher development risks. This creates unique challenges that have led to innovative funding models and collaborative approaches made specifically for people living with rare diseases.
Government agencies provide substantial foundational support for rare disease research through targeted programs and initiatives. The U.S Food and Drug Administration (FDA) operates several grant programs specifically designed for rare conditions, including the Natural History Studies Grants Program, which supports research that advances medical product development by identifying patient populations and establishing biomarkers.
The FDA's Rare Neurodegenerative Disease Grants Program represents a focused approach to funding conditions like ALS and other rare neurological disorders. These grants help cover research and development costs for interventions where traditional funding sources may be insufficient due to small patient populations.

The National Center for Advancing Translational Sciences (NCATS), part of the National Institutes of Health (NIH), supports rare disease research through extramural programs that bridge the gap between laboratory discoveries and clinical applications. NCATS funding often focuses on developing research tools and methodologies that can be applied across multiple rare diseases, maximizing the impact of federal investment.
The National Institutes of Health (NIH) also supports collaborative initiatives through programs like the Centers of Excellence in Regulatory Science and Innovation, which strengthen partnerships between academic institutions and regulatory agencies. These programs provide cooperative agreements that support long-term research initiatives and help establish the scientific infrastructure needed for rare disease studies.
Patient advocacy organizations serve as critical funding sources, often representing the only financial support available for studying specific rare diseases, especially ultra-rare ones that get no attention from government or industry. The National Organization for Rare Disorders (NORD) operates a competitive grant program that provides seed funding for translational and clinical studies. Most of the money comes directly from patients, families, and patient advocacy organizations themselves.
NORD's grant program requires a minimum of $35,000 per project, with $55,000 recommended for more comprehensive studies. Donors can direct their contributions toward research on specific disorders, creating a direct link between affected communities and scientific progress. The organization follows NIH-style review procedures, ensuring scientific rigor while maintaining focus on patient priorities.
These community-driven grants frequently generate preliminary data that researchers can later use to secure larger funding from government agencies or pharmaceutical companies. In many cases, they help fill critical gaps left by traditional funding sources.
Rare diseases don’t respect borders. So cross-country collaboration has become essential for rare disease research, leading to the development of international funding mechanisms. The European Rare Disease Research Alliance (ERDERA) exemplifies this approach, pooling resources from regional and national agencies, the European Commission, private sector organizations, and philanthropic foundations.
The European Rare Disease Research Alliance (ERDERA) operates three distinct funding mechanisms designed to address different research needs. Joint Transnational Calls support interdisciplinary research projects that require cross-country collaboration, bringing together expertise from multiple countries to address identified research gaps. Clinical Trials Calls specifically fund controlled human studies focused on establishing therapeutic safety and effectiveness.
The organization's Networking Support Scheme helps share knowledge and collaboration between research groups, recognizing that rare disease research benefits significantly from coordinated international efforts. This pooled funding approach allows smaller countries with limited resources to participate in large-scale research initiatives that would be impossible to support individually.
Specialized research networks receive dedicated funding to support the infrastructure needed for rare disease studies. The Rare Diseases Clinical Research Network (RDCRN) in the U.S operates 21 consortia that, along with affiliated patient advocacy groups, offer various fellowship and grant funding opportunities.
These networks provide early career grants, biomarker development programs, clinical research training scholarships, and young investigator funding across diverse rare disease conditions. The network model creates sustainable research communities that can pursue long-term studies and maintain institutional knowledge about specific conditions.
Funding for research infrastructure includes support for patient registries, natural history studies, and biobanking initiatives. These resources make future research easier and more efficient by creating standardized data collection methods and sample repositories that researchers can access for multiple studies.
Private foundations contribute significant funding for rare disease research, often focusing on specific therapeutic areas or research approaches. These organizations typically operate with greater flexibility than government agencies, allowing them to fund innovative or high-risk projects that might not receive traditional grant support.
Philanthropic funding often supports early-stage research that generates proof-of-concept data for novel therapeutic approaches. Foundation grants frequently bridge the gap between basic research discoveries and the applied research necessary for treatment development.
Some foundations focus specifically on funding research for diseases that affect children, recognizing the unique challenges and urgency associated with pediatric rare conditions. Others target diseases with particular research challenges, such as those affecting the nervous system or involving genetic mutations.
Research funding strategy in the rare disease field relies heavily on systematic gap analysis to identify the most pressing needs within the community. Funding organizations gather recommendations from advisory and governing bodies to ensure resources target areas with the greatest unmet medical needs.
This strategic approach recognizes that funding decisions must balance scientific opportunity with patient impact. Priority setting involves evaluating factors such as disease severity, lack of existing treatments, research feasibility, and the potential for developing effective interventions.
Gap analysis also identifies opportunities for collaborative funding where multiple organizations can pool resources to support larger studies than any single funder could manage alone. This coordination helps avoid duplication of effort and ensures comprehensive coverage of research priorities.
The funding landscape for rare diseases faces several structural challenges that affect research progress. Small patient populations make traditional clinical trials expensive and difficult, requiring innovative study designs that may not fit standard funding criteria. Limited commercial viability means pharmaceutical companies often cannot justify the investment required for drug development.
Regulatory requirements designed for common diseases may not be appropriate for rare conditions, creating additional costs and complexity that funding must address. The need for specialized expertise often means research can only be conducted at a few institutions worldwide, requiring funding models that support international collaboration.
The geographic distribution of patients presents another funding challenge, as research often requires enrolling people from different countries to gather a sufficient number of participants. This requires funding systems capable of supporting international research at multiple sites, despite the complex coordination involved.
The rare disease funding landscape continues to evolve with new models emerging to address persistent challenges. Collaborative funding initiatives are expanding to include a broader range of stakeholders, bringing together government agencies, patient organizations, pharmaceutical companies, and philanthropic foundations in shared funding arrangements.
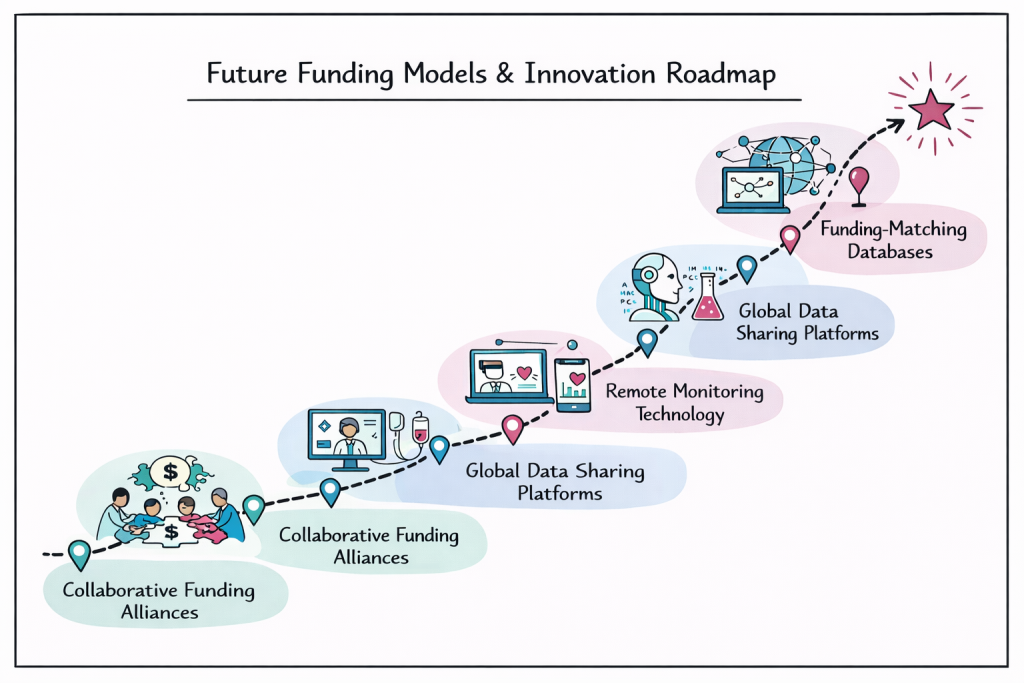
Technology-enabled research platforms are creating new funding opportunities by reducing the cost and complexity of conducting rare disease studies. Virtual clinical trials and remote monitoring capabilities allow funders to support studies that would have been impossible or prohibitively expensive using traditional methods.
Data sharing initiatives are maximizing the value of funding investments by ensuring research results can be used by multiple research groups. These collaborative approaches help stretch limited funding resources while accelerating the pace of discovery across the rare disease field.
The development of specialized funding databases and matching services helps connect researchers with appropriate funding opportunities, reducing the time and effort required to identify potential support sources. These tools recognize that successful rare disease research depends on an efficient connection between scientific expertise and available resources.
Rare disease research funding remains limited compared to common diseases, but innovative collaborative models and community-driven initiatives are steadily advancing progress. By combining government support, patient advocacy, international collaboration, and philanthropic investment, the rare disease community continues to push boundaries, bringing hope to millions of patients worldwide.