Explore how endpoints are defined in rare disease clinical trials, the challenges of small populations, and the growing role of patient-reported outcomes in regulatory decisions.
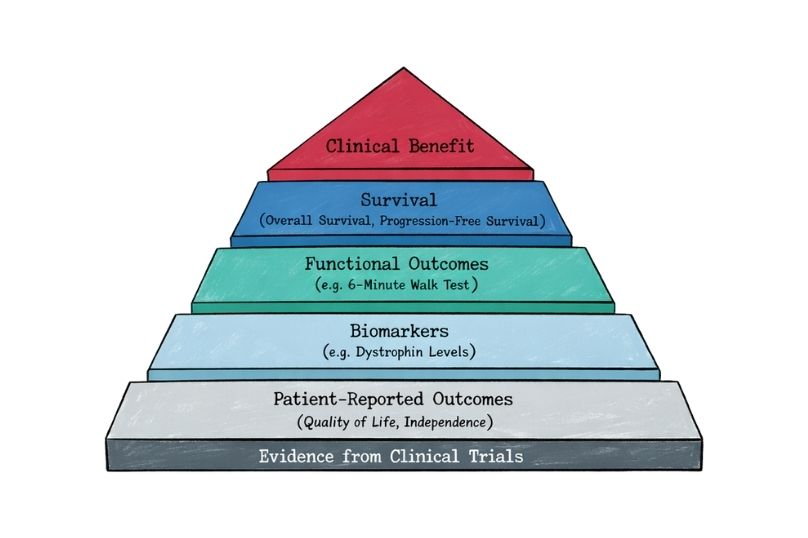 Endpoints in Rare Disease Clinical Trials
Endpoints in Rare Disease Clinical Trials
Explore how endpoints are defined in rare disease clinical trials, the challenges of small populations, and the growing role of patient-reported outcomes in regulatory decisions.
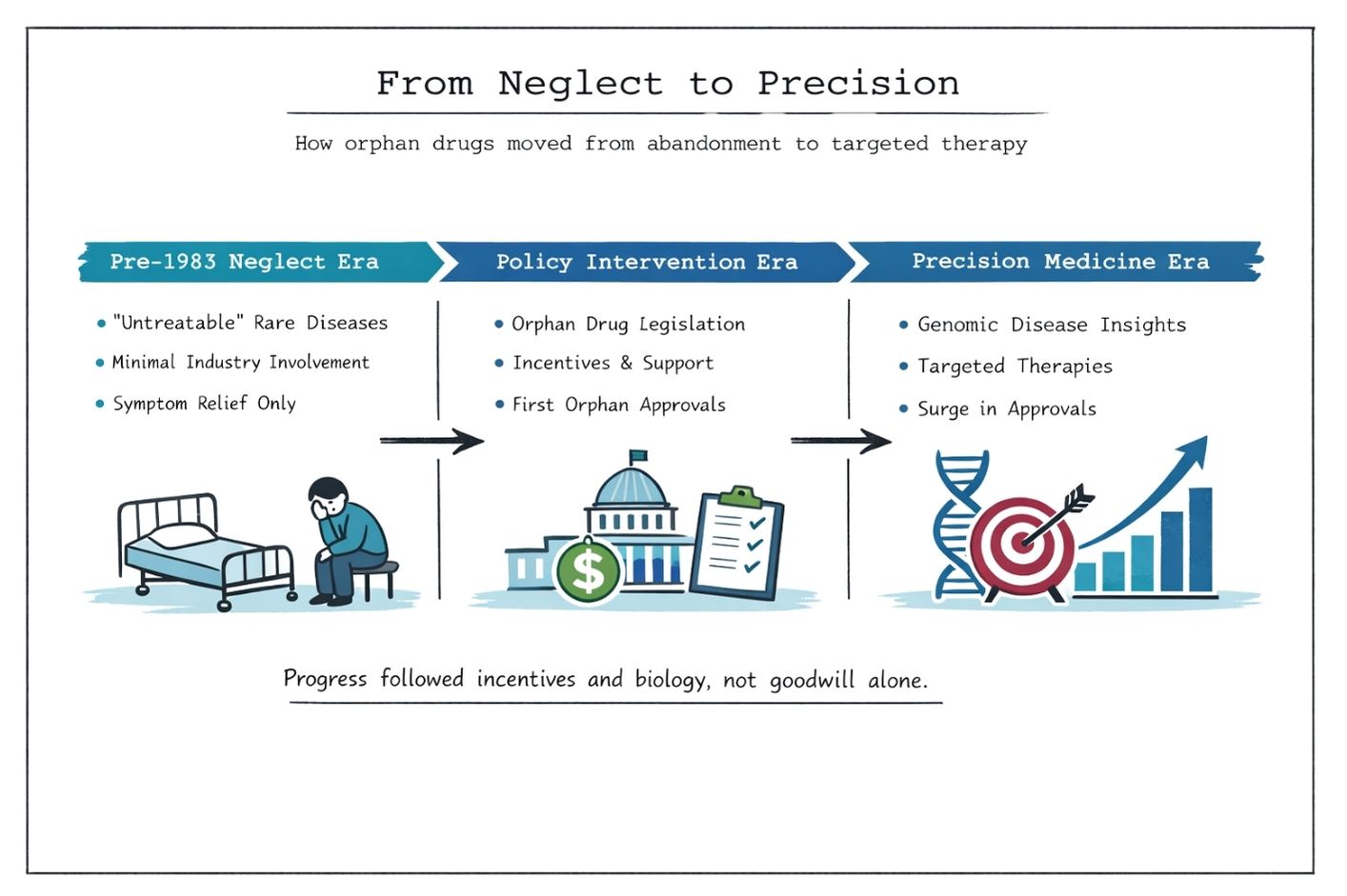 Orphan Drugs: From Neglect to Precision Medicine
Orphan Drugs: From Neglect to Precision Medicine
Although orphan drug legislation and genomic science have transformed the therapeutic landscape for rare diseases, the path from scientific discovery to patient access remains complex and uncertain.