Rare disease research is transformative, yet traditional investment models struggle to support innovation in this field. Unlike common diseases with large patient populations, rare diseases face unique economic challenges. Addressing these challenges often requires innovative funding approaches from both industry and public institutions. Thus, the financial landscape for rare disease research and care is shaped by a complex network of funding sources, each with distinct roles and objectives.
Developing treatments for rare diseases presents significant financial challenges. Rare diseases collectively impact an estimated 300 million people globally. However, individual conditions often involve small patient populations, typically in the hundreds or low thousands worldwide. This makes it difficult for traditional pharmaceutical models to justify the high costs of research and development.
The average cost of bringing a new drug to market exceeds $2.6 billion. In the context of rare diseases, where potential revenue is limited, this creates a challenging economic environment that can slow progress and restrict therapeutic development.
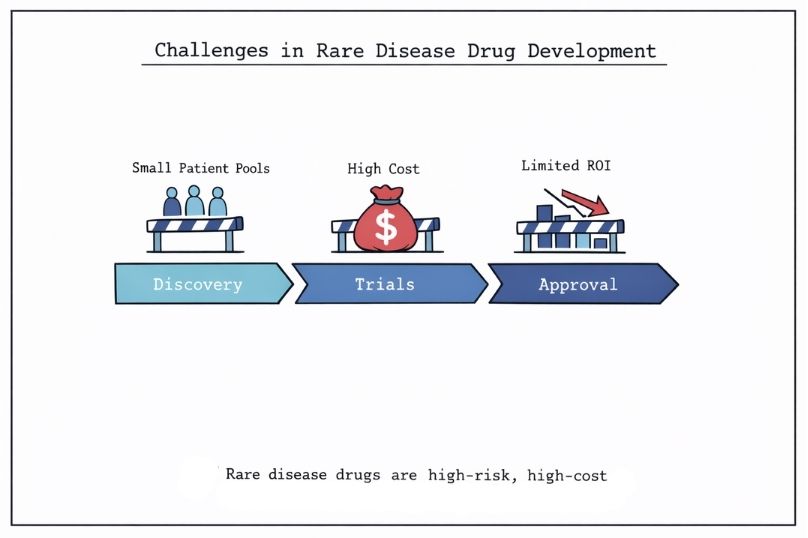
In response, a range of innovative funding approaches has emerged to bridge the gap between commercial viability and medical need. These include contributions from pharmaceutical companies, biotechnology firms, venture capital, private foundations, and hybrid models that combine financial investment with philanthropic objectives.
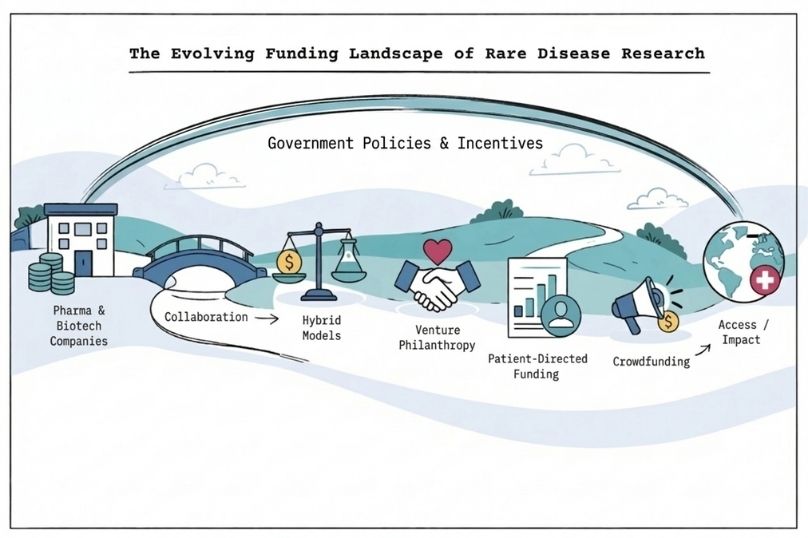
Pharmaceutical and biotechnology companies play a central role in funding rare disease research and development through a range of strategic approaches. Many large pharmaceutical companies have established dedicated rare disease divisions that operate with different financial expectations than traditional drug development programmes. These divisions often accommodate longer development timelines and lower return-on-investment thresholds in exchange for regulatory incentives such as market exclusivity and orphan drug designation.
Biotechnology companies, particularly smaller firms, often focus exclusively on rare diseases. They typically secure funding through venture capital, strategic partnerships with larger pharmaceutical companies, or licensing agreements. This specialised focus allows them to develop deep expertise in specific therapeutic areas, making them attractive acquisition partners or acquisition targets for larger companies seeking to expand their rare disease portfolios.
Genzyme, later acquired by Sanofi in 2011, is a leading example of how biotech innovation can scale through industry integration. Its flagship therapy, Cerezyme (imiglucerase) for Gaucher disease, became one of the earliest and most successful orphan drugs. Genzyme’s focused research and patient-centred approach highlight how specialised expertise can drive innovation in rare disease treatment.
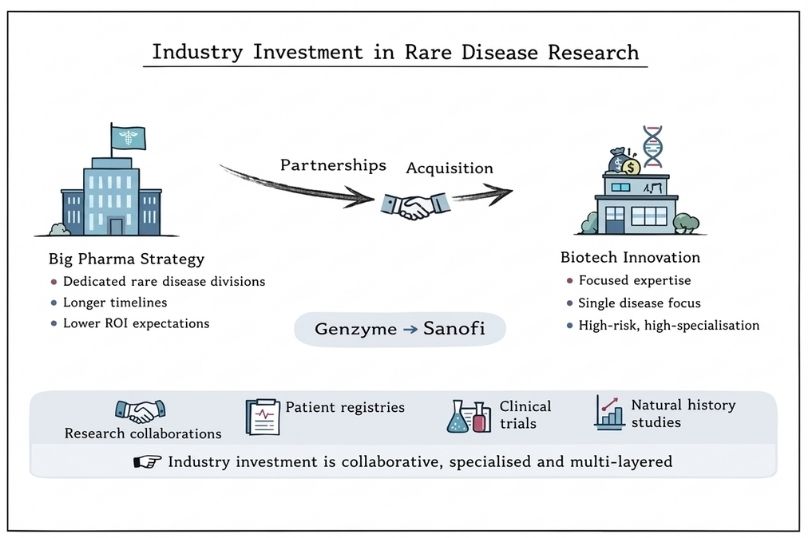
Industry funding mechanisms extend beyond direct investment in drug development. These may include collaborative research agreements with academic institutions, support for patient registries, and funding for clinical trials. Companies may also invest in natural history studies, which provide critical insights into disease progression and help identify potential therapeutic targets.
Private foundations have emerged as important intermediaries between traditional philanthropy and commercial investment in rare disease research. Between 2002 and 2017, philanthropic investments in life sciences increased five-fold to reach $2.6 billion in the United States, reflecting growing recognition of this funding model's potential.
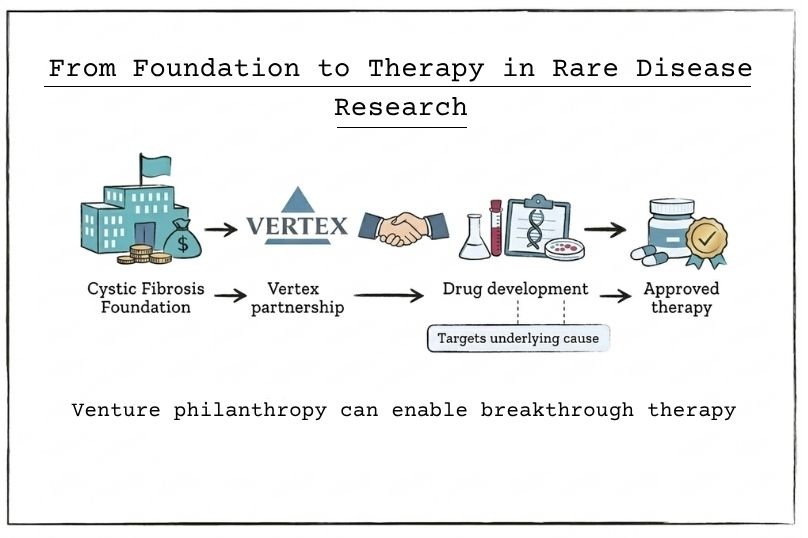
Many foundations now adopt venture philanthropy approaches, using tools such as programme-related investments (PRIs). These mechanisms enable foundations to provide loans, take equity positions, or enter licensing agreements that generate financial returns while advancing their charitable objectives.
A widely cited example is the Cystic Fibrosis Foundation, which invested in the development of targeted therapies through a partnership with Vertex Pharmaceuticals. This approach contributed to treatments that address the underlying causes of cystic fibrosis, rather than focusing solely on symptom management.
Some foundations have created their own for-profit life sciences subsidiaries specifically focused on rare disease research. This structure requires careful legal and operational design to maintain nonprofit status while pursuing commercial drug development, but it allows foundations to retain greater control over research priorities and development timelines.
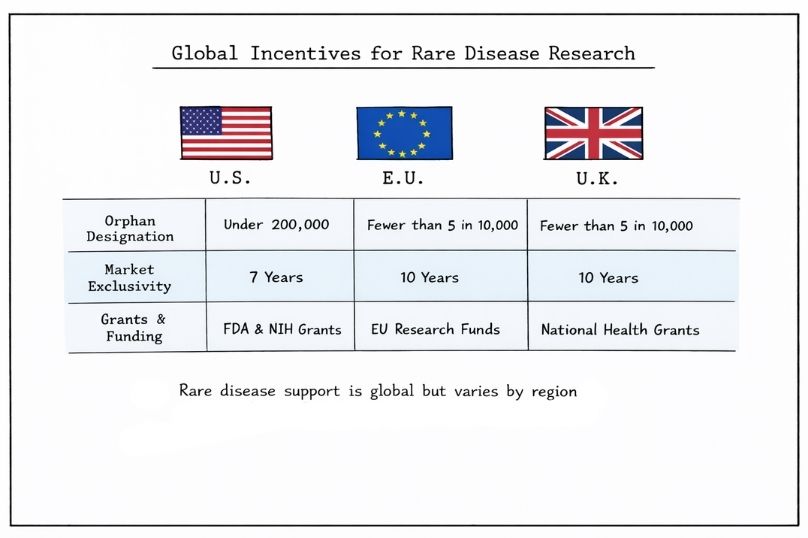
Government incentives are a key driver of rare disease research and development, helping to offset the financial risks associated with small patient populations. In the United States, the Orphan Drug Act of 1983 established a framework of financial and regulatory incentives for therapies targeting diseases affecting fewer than 200,000 individuals. In the European Union, orphan designation is granted for conditions affecting fewer than 5 in 10,000 people, while the United Kingdom continues to apply similar criteria post-Brexit through its national regulatory framework.
Key government funding and incentive mechanisms include
Orphan designation programmes across the US, EU, and UK provide benefits such as protocol assistance, reduced regulatory fees, and market exclusivity. In the US, approved therapies receive seven years of exclusivity, while in the EU and UK, this period typically extends to ten years.
Government agencies support rare disease research through grants spanning early-stage discovery to clinical development. In the US, this includes programmes such as the Food and Drug Administration (FDA)’s Orphan Products Grants Programme and funding from the National Institutes of Health (NIH). In Europe, initiatives such as European Rare Diseases Research Alliance (ERDERA) and national funding bodies provide similar support for collaborative and translational research.
In the United States, developers may claim tax credits on qualified clinical research expenses. Comparable financial incentives, including grants and co-funding schemes, are available across EU member states and the UK to reduce development risk.
Programmes such as the FDA’s Rare Disease Endpoint Advancement Programme, alongside initiatives led by the European Medicines Agency (EMA), support the development of novel clinical endpoints and trial designs. These efforts aim to accelerate development timelines while maintaining regulatory standards.
Collectively, these incentives play a critical role in making rare disease drug development more viable, bridging the gap between scientific opportunity and commercial feasibility across global healthcare systems.
Patient advocacy organisations and disease-specific foundations represent another important source of funding in rare disease research, particularly for ultra-rare conditions affecting very small patient populations. In many cases, these organisations provide essential support where limited commercial or public funding is available.
In the United States, the National Organisation for Rare Disorders (NORD) plays a key role in coordinating patient-driven funding through research grant programmes supported by individual donors, families and advocacy groups. These initiatives help direct resources toward specific diseases, often reflecting the priorities of patient communities.
NORD's funding model requires minimum thresholds of $35,000 with $55,000 recommended before issuing research proposals, ensuring sufficient resources to conduct meaningful studies. Donors can make direct contributions to specific diseases, creating a direct connection between patient communities and research funding.
In Europe, EURORDIS represents a network of over 900 patient organisations. It integrates patient perspectives into research and regulatory frameworks, including contributions to clinical trial design and European reference networks. In the United Kingdom, Genetic Alliance UK coordinates more than 200 organisations and contributes to national strategies such as the UK Rare Diseases Framework, supporting patient registries, data sharing initiatives, and research prioritisation.
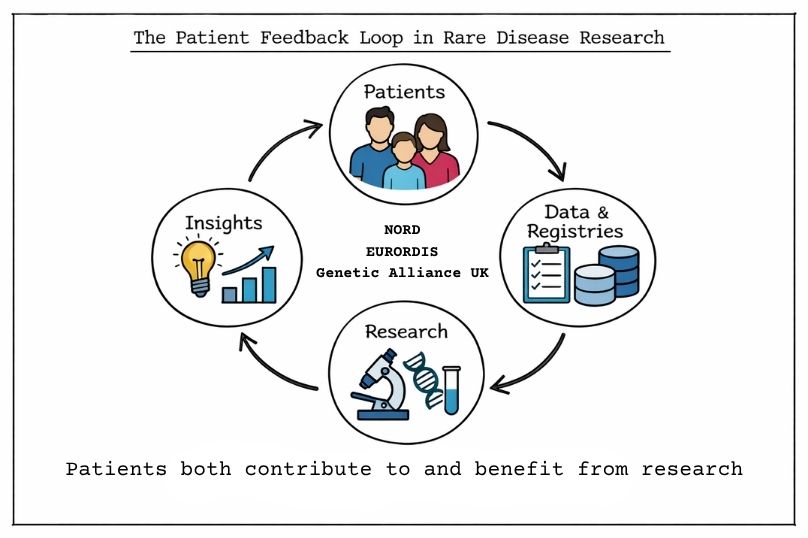
Patient organisations often serve as intermediaries within the rare disease ecosystem. They translate complex medical research needs into accessible fundraising campaigns, while also maintaining relationships with researchers, clinicians and funding partners. Beyond financial contributions, these organisations support a range of activities that help advance research like the development of patient registries, natural history studies, and clinical trial recruitment.
Collaborative infrastructure and support networks play an important role in strengthening rare disease research, particularly in areas where resources and expertise are limited. By enabling coordination across stakeholders, these networks help address the fragmentation that often characterises rare disease research.
Organisations such as the RTW Foundation provide specialised expertise to ultra-rare disease initiatives, offering operational support and strategic guidance for drug discovery projects. Similarly, the Rare Diseases Clinical Research Network (RDCRN) maintains 21 consortia that facilitate multi-centre collaboration, as well as fellowship and grant opportunities through affiliated patient advocacy groups. These structures enable shared infrastructure, standardised protocols, and coordinated research efforts across institutions.
Translational award programmes further strengthen this ecosystem by combining funding with drug development expertise. Initiatives such as the Oxford-Harrington Rare Disease Scholar Award support the progression of academic discoveries into clinical therapies. By integrating financial support with industry knowledge, these programmes help bridge the gap between early-stage research and therapeutic development.
The Critical Path Institute exemplifies this collaborative approach through its programmes. Its Rare Disease Outcome Assessment Consortium brings together stakeholders across industry, academia, regulators, and patient groups to develop standardised outcome measures, strengthening clinical trial design and evaluation. Complementing this, the Accelerator Data and Analytics Programme supports secure data sharing and advanced analytics, enabling more coordinated and data-driven collaboration across the rare disease research ecosystem.
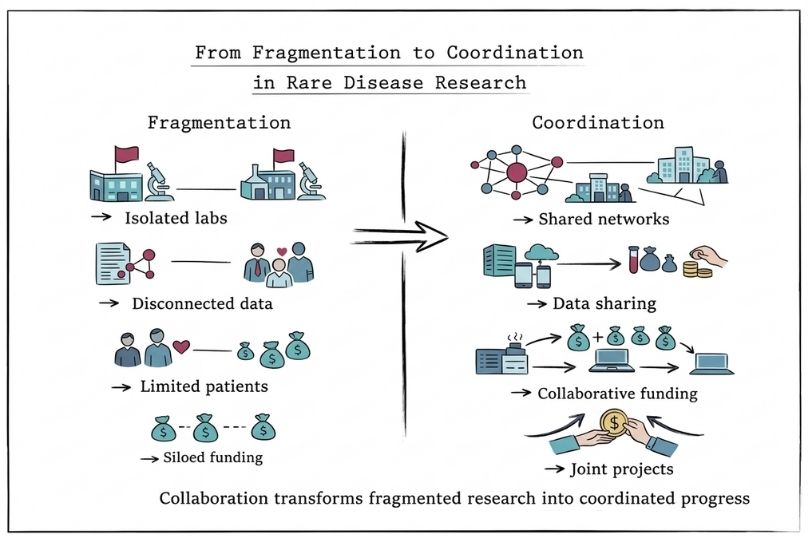
These public-private partnerships leverage expertise and resources from multiple stakeholders while sharing risks and costs.
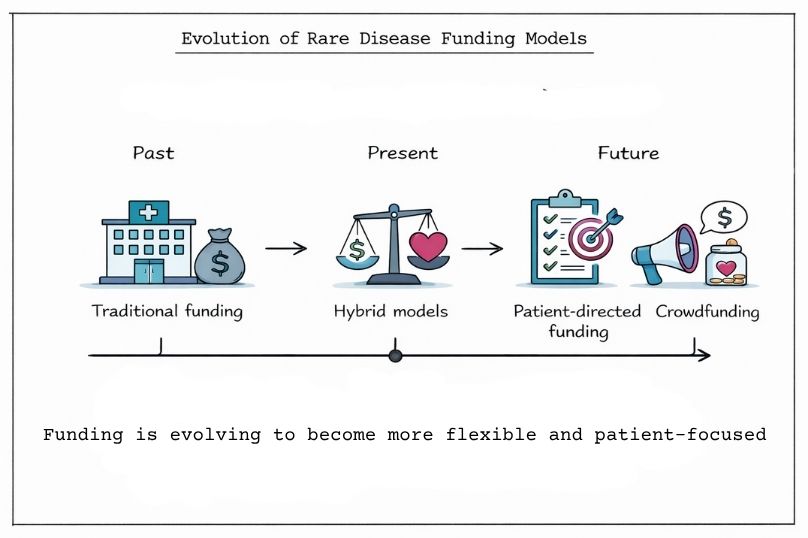
The rare disease funding landscape continues to evolve, with new hybrid models that combine commercial and philanthropic elements. Innovative models such as social impact bonds, milestone-based payments, and tiered pricing structures are emerging as mechanisms to align financial incentives with patient outcomes.
In parallel, some organisations are adopting patient-directed funding approaches. In these models, patient advocacy groups may directly commission specific research activities, rather than providing unrestricted grants. This can enable greater alignment between research priorities and patient needs, while maintaining scientific and regulatory standards.
Crowdfunding platforms specifically designed for rare disease research are also gaining traction, allowing individual patients and families to directly support research into their specific conditions. Although typically smaller in scale compared to traditional funding sources, they can provide an important avenue of support for ultra-rare diseases that may otherwise receive limited attention.
The sustainability of rare disease funding depends on continued collaboration between industry, government, and patient communities. Progress in one rare condition often generates insights that can be applied more broadly, creating spillover benefits that justify broader investment.
Regulatory agencies continue to refine incentive frameworks to encourage rare disease research while ensuring patient safety and treatment efficacy. The FDA's accelerated approval pathways and breakthrough therapy designations provide additional incentives for companies willing to invest in rare disease development.
There is also growing recognition that rare disease research contributes to advances in fields such as precision medicine and gene therapy. As these technologies evolve, the traditional boundaries between rare and more common conditions may become less distinct, potentially reshaping funding models and development strategies.
Looking ahead, sustaining progress will require a balanced and collaborative funding ecosystem, supported by new approaches to address challenges in drug discovery and development. Ultimately, progress in rare disease research must translate beyond innovation into earlier diagnosis and equitable access to care, reducing the diagnostic odyssey faced by millions of patients worldwide.