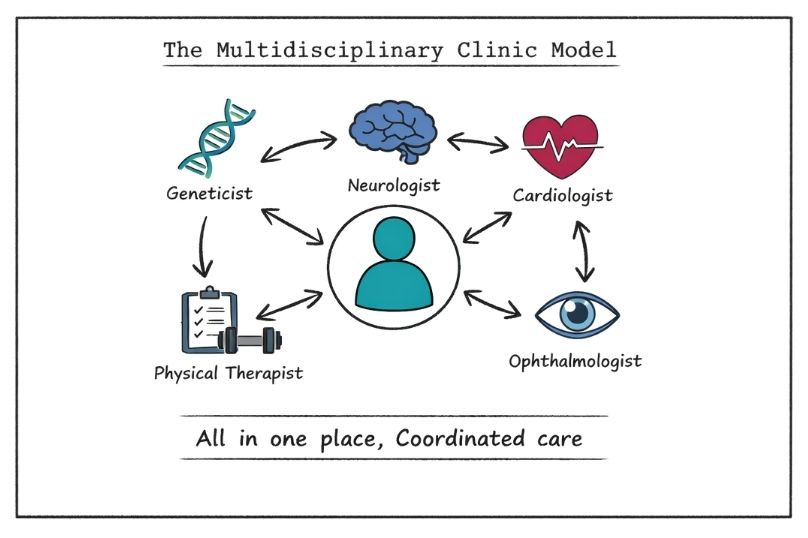
Multidisciplinary clinics for rare diseases unite specialists from different fields to collaborate on diagnosis and treatment, helping patients receive faster answers, coordinated care, and access to advanced therapies."
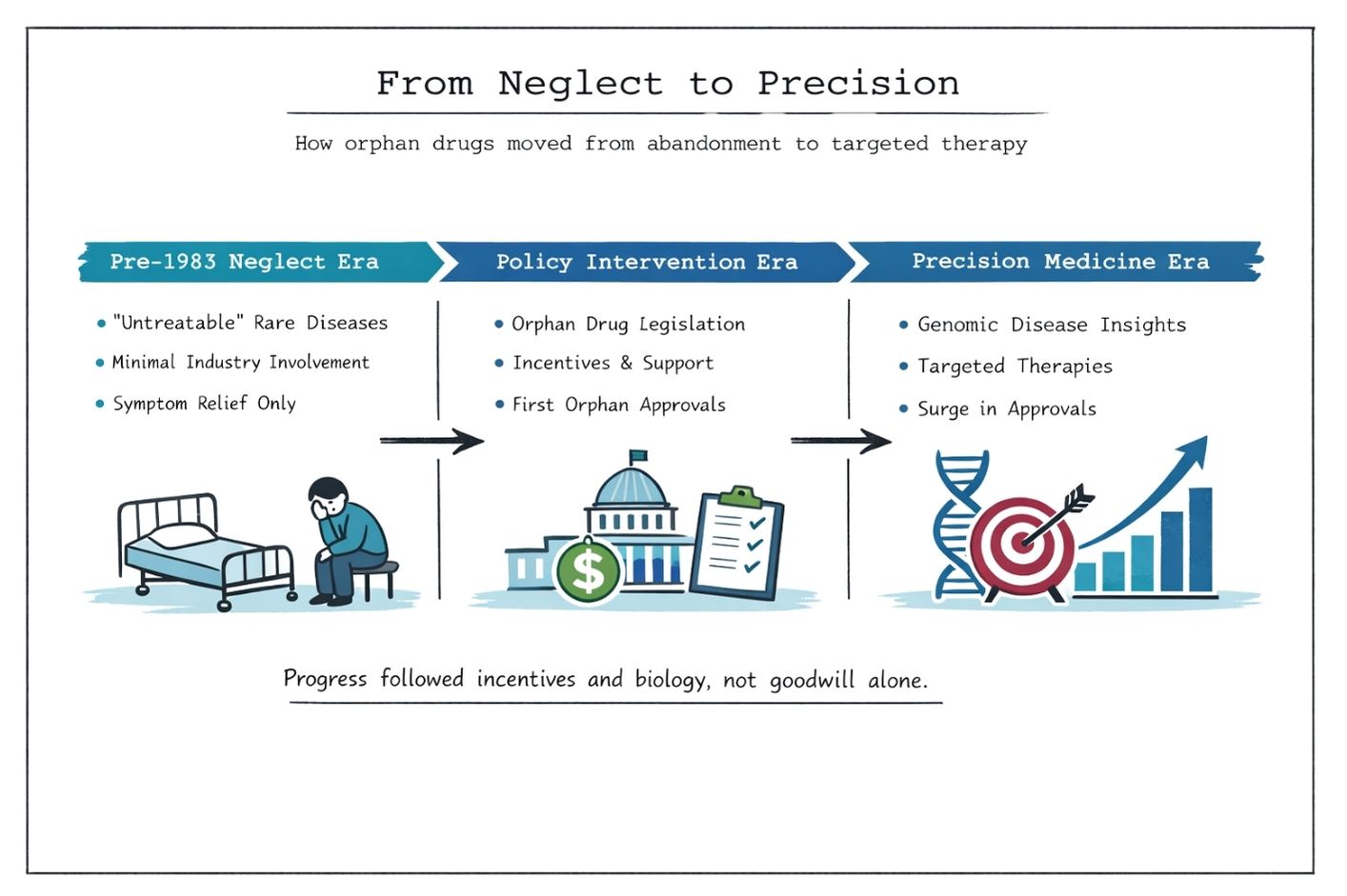
Articles exploring how orphan drug designation works, why rare disease medicines can be expensive, how access decisions are made, and what patients can realistically expect from new therapies.
We also report on treatment approvals, clinical trial results, safety updates, and changes in availability across different countries. Alongside news and analysis, we signpost reliable sources of information, patient organisations, and specialist centres where people may find further guidance or support.
The aim is to provide clear, evidence-based information about treatments without exaggerating benefits or minimising uncertainty.